Advertisements
Advertisements
प्रश्न
The increasing order of reduction of alkyl halides with zinc and dilute HCl is ______.
विकल्प
R – Cl < R – I < R – Br
R – Cl < R – Br < R – I
R – I < R – Br < R – Cl
R – Br < R – I < R – Cl
उत्तर
The increasing order of reduction of alkyl halides with zinc and dilute HCl is R – Cl < R – Br < R – I.
Explanation:
The reactivity of reduction bf alkyl halides with Zn/HCl increases as the strength of the C – X bond decreases, i.e., R – Cl < R – Bf < R – I.
APPEARS IN
संबंधित प्रश्न
Write the structures and names of products obtained in the reactions of sodium with a mixture of 1-iodo-2-methylpropane and 2-iodopropane.
An unsaturated hydrocarbon ‘A’ adds two molecules of H2 and on reductive ozonolysis gives butane-1,4-dial, ethanal and propanone. Give the structure of ‘A’, write its IUPAC name and explain the reactions involved.
Identify a CORRECT statement about the preparation of alkanes from the following:
An alkyl halide by formation of its Grignard reagent and heating with water gives propane. What is the original alkyl halide?
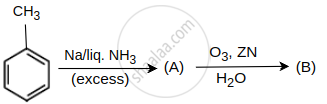
Identify products A and B.

Which of the following is Lindlar's catalyst?
The number of nitrogen atoms in a semicarbazone molecule of acetone is ______.
\[\ce{X + 3NH3 -> Y ->[H^+/H2O]}\]
H2N–CH2–COOH, compound X is:
\[\ce{X <-[red P][HI] CH3COOH ->[LiAlH4] Y.}\]
What does Not true for X and Y?
In the given reaction final product(s) will be: