Advertisements
Advertisements
प्रश्न
An unsaturated hydrocarbon ‘A’ adds two molecules of H2 and on reductive ozonolysis gives butane-1,4-dial, ethanal and propanone. Give the structure of ‘A’, write its IUPAC name and explain the reactions involved.
उत्तर
Two molecules of hydrogen add on ‘A’ this shows that ‘A’ is either an alkadiene or an alkyne.
On reductive ozonolysis ‘A’ gives three fragments, one of which is dialdehyde. Hence, the molecule has broken down at two sites. Therefore, ‘A’ has two double bonds. It gives the following three fragments:
OHC – CH2 – CH2 – CHO, CH3CHO and CH3 – CO – CH3
Hence, its structure as deduced from the three fragments must be
\[\begin{array}{cc}
\ce{CH3 - CH = CH - CH2 - CH2 - CH = C - CH3}\\
\phantom{.................................}|\\
\phantom{...................................}\ce{CH3}
\end{array}\]
(A)
Reactions
APPEARS IN
संबंधित प्रश्न
The increasing order of reduction of alkyl halides with zinc and dilute HCl is ______.
Match the hydrocarbons in Column I with the boiling points given in Column II.
| Column I | Column II |
| (i) n-Pentane | (a) 282.5 K |
| (ii) iso-Pentane | (b) 309 K |
| (iii) neo-Pentane | (c) 301 K |
896 mL vapour of a hydrocarbon ‘A’ having carbon 87.80% and hydrogen 12.19% weighs 3.28 g at STP. Hydrogenation of ‘A’ gives 2-methylpentane. Also ‘A’ on hydration in the presence of H2SO4 and HgSO4 gives a ketone ‘B’ having molecular formula C6H12O. The ketone ‘B’ gives a positive iodoform test. Find the structure of ‘A’ and give the reactions involved.
Identify a CORRECT statement about the preparation of alkanes from the following:
An alkyl halide by formation of its Grignard reagent and heating with water gives propane. What is the original alkyl halide?
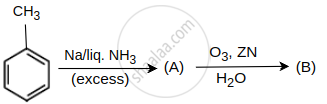
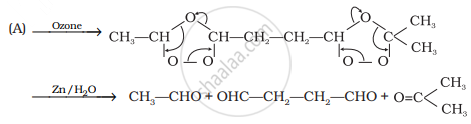
Identify products A and B.

Which of the following is Lindlar's catalyst?
The number of nitrogen atoms in a semicarbazone molecule of acetone is ______.
\[\ce{X <-[red P][HI] CH3COOH ->[LiAlH4] Y.}\]
What does Not true for X and Y?
In the given reaction final product(s) will be: