Advertisements
Advertisements
प्रश्न
The nuclei of isotopes of a given element contain the same number of ______.
विकल्प
Neutrons
Neutrinos
Positrons
Protons
MCQ
रिक्त स्थान भरें
उत्तर
The nuclei of isotopes of a given element contain the same number of protons.
Explanation:
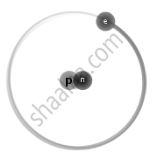
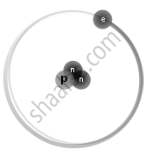
Isotopes are different neutron number variants of the same chemical element. Each atom in all isotopes of a specific element has the same number of protons. Example:
 |
 |
 |
| `"_1^1H` Protium |
`"_1^2H` Deuterium |
`"_1^3H` Tritium |
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
