Advertisements
Advertisements
प्रश्न
The number of neutrons present in 73Li is ______.
उत्तर
The number of neutrons present in 73Li is 4
APPEARS IN
संबंधित प्रश्न
The mass of an electron is about `1/2000` times that of proton.
Which subatomic particle is not present in an ordinary hydrogen atom?
Name the subatomic particle whose relative charge is : 0
Fill in the blank of the following statement :
If the nucleus of an atom has atomic number 17, mass number 37 and there are 17 electrons outside the nucleus, the number of neutrons in it is __________.
Explain in brief the experimental proof which led to the discovery of –
Neutrons
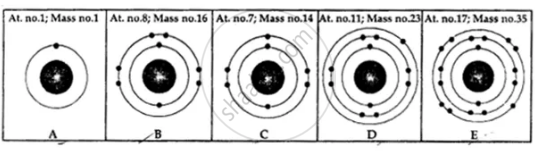
State the number of neutrons in each of the atoms A to E. Also state which of the atoms A to E is a metal.
Calculate the number of neutrons, protons and electrons:
- atomic number 3 and mass number 7
- atomic number 92 and mass number 238.
James Chadwick discovered the fundamental particle called proton.
What is the charge of a neutron?
Which subatomic particles have no charge?
