Advertisements
Advertisements
प्रश्न
The number of terminal oxygen atoms present in the product B obtained from the following reactions is:
\[\ce{FeCr2O4 + Na2CO3 + O2 -> A + Fe2O3 + CO2}\]
\[\ce{A + H^+ -> B + H2O + Na^+}\]
विकल्प
2
4
6
8
MCQ
उत्तर
6
Explanation:
\[\ce{FeCr2O4 + Na2CO3 + O2 -> \underset{(A)}{Na2CrO4} + Fe2O3 + CO2}\]
\[\ce{\underset{(A)}{Na2CrO4} + H^+ -> \underset{(B)}{Na2Cr2O7} + Na^+ + H2O}\]
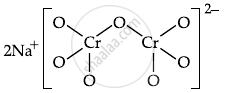
The number of terminal oxygen atoms present in the product B is 6.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
