Advertisements
Advertisements
प्रश्न
The presence of nitro group (−NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.
उत्तर
The presence of nitro groups (−NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions because nitro groups (−NO2) at o/p positions withdraw the electron density from the benzene ring facilitating the attack of the nucleophile. The negative charge in the carbanion formed, at ortho and para positions with respect to halogen atom, is stabilised through resonance and the presence of nitro groups (−NO2), respectively.
APPEARS IN
संबंधित प्रश्न
What happens when \[\ce{CH3 - Br}\] is treated with KCN?
Write the final product(s) in each of the following reactions:
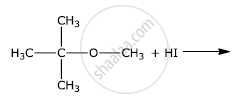
How the following conversion can be carried out?
Chlorobenzene to p-nitrophenol
Write the product formed on reaction of D-glucose with Br2 water.
What is Grignard reagent?
Out of (CH3)3 C-Br and (CH3)3 C-I, which one is more reactive towards SN1 and why?
Which of the following compounds will give racemic mixture on nucleophilic substitution by \[\ce{OH-}\] ion?
(a) \[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - Br}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
(b) \[\begin{array}{cc}
\phantom{..}\ce{Br}\\
\phantom{}|\\
\phantom{}\ce{CH3 - C - CH3}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
(c) \[\begin{array}{cc}
\phantom{....}\ce{CH3 - CH - CH2Br}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 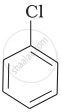 |
| (b) | 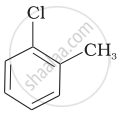 |
| (c) | 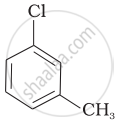 |
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 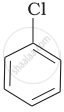 |
| (b) | 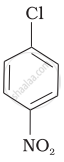 |
| (c) | 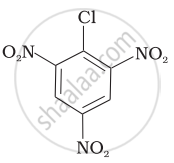 |
Allyl chloride is hydrolysed more readily than n-propyl chloride. Why?
