Advertisements
Advertisements
प्रश्न
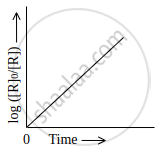
The slope in the plot of `log ["R"]_0/(["R"])` Vs. time for a first-order reaction is ______.
विकल्प
`(+"k")/2.303`
+k
`(-"k")/2.303`
−k
उत्तर
The slope in the plot of `log ["R"]_0/(["R"])` Vs. time for a first-order reaction is `bbunderline((+"k")/2.303)`.
Explanation:
For the reaction, \[\ce{R -> Products}\]
The integrated first-order rate equation is
log [R] = `(−"kt")/2.303 + log ["R"_0]`
or `log (["R"_0])/(["R"]) = ("kt")/2.303`
Comparing the above equation with a straight-line equation y = mx + c
∴ y = `log (["R"_0])/(["R"])`, x = t, slope = `"k"/2.303`
APPEARS IN
संबंधित प्रश्न
In a pseudo first order hydrolysis of ester in water, the following results were obtained:
| t/s | 0 | 30 | 60 | 90 |
| [A]/mol L−1 | 0.55 | 0.31 | 0.17 | 0.085 |
Calculate the average rate of reaction between the time interval 30 to 60 seconds.
A first order reaction takes 20 minutes for 25% decomposition. Calculate the time when 75% of the reaction will be completed.
(Given : log = 2 = 0·3010, log 3 = 0·4771, log 4 = 0·6021)
A first order reaction is 50% complete in 25 minutes. Calculate the time for 80% completion of the reaction.
Straight line graph for first order reaction is obtained between ____________.
Which of the following graphs is correct for a first order reaction?

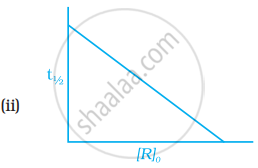
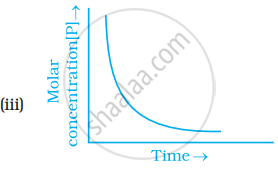
In the presence of acid, the initial concentration of cane sugar was reduced from 0.2 M to 0.1 Min 5 hours and to 0.05 Min 10 hours. The reaction must be of?
The rate constant of a first order reaction is 6.9 × 10–3s–1. How much time will it take to reduce the initial concentration to its 1/8th value?
First order reaction is 50% complete in 1.26 × 1014s. How much time could it take for 100% completion?
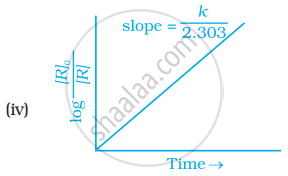
Observe the graph shown in figure and answer the following questions:
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
The rate constant for the reaction:
\[\ce{2N2O_{(s)} ->2N2O4_{(g)}}\] is 4.98 × 10-4 s-1.
The order of the reaction is ______.
