Advertisements
Advertisements
प्रश्न
Two neutral particles are kept 1 m apart. Suppose by some mechanism some charge is transferred from one particle to the other and the electric potential energy lost is completely converted into a photon. Calculate the longest and the next smaller wavelength of the photon possible.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
उत्तर
Given:-
Distance between the two neutral particles, r = 1 m
Electric potential energy,
`E_1 = (kq^2)/r = kq^2`
where `K = 1/(4pi∈_0)`
Energy of photon,
`E_2 = (hc)/λ`,
where λ = wavelength of light
h = Planck's constant
c = speed of light
Here, E1 = E2
`therefore kq^2 = (hc)/λ`
`⇒ λ = (hc)/(kq^2)`
For wavelength, λ, to be maximum, charge q should be minimum.
`q = e = 1.6 xx 10^-19 "C"`
Maximum wavelength,
`λ = (hc)/(kq^2)`
`= (6.63 xx 3 xx 10^-34 xx 10^8)/(9 xx 10^2 xx (1.6)^2 xx 10^-38)`
`= 0.863 xx 10^3 = 863 "m"`
Next smaller wave length,
`λ = (6.63 xx 3 xx 10^-34 xx 10^8)/(9 xx 10^4 xx 4 xx (1.6)^2 xx 10^-38)`
`= 863/4`
`= 215.74 "m"`
APPEARS IN
संबंधित प्रश्न
The threshold frequency for a certain metal is 3.3 × 1014 Hz. If light of frequency 8.2 × 1014 Hz is incident on the metal, predict the cutoff voltage for the photoelectric emission.
Light of frequency 7.21 × 1014 Hz is incident on a metal surface. Electrons with a maximum speed of 6.0 × 105 m/s are ejected from the surface. What is the threshold frequency for photoemission of electrons?
Is the formula you employ in (a) valid for calculating radius of the path of a 20 MeV electron beam? If not, in what way is it modified?
What is so special about the combination e/m? Why do we not simply talk of e and m separately?
If light of wavelength 412.5 nm is incident on each of the metals given below, which ones will show photoelectric emission and why?
| Metal | Work Function (eV) |
| Na | 1.92 |
| K | 2.15 |
| Ca | 3.20 |
| Mo | 4.17 |
Define the term "cut off frequency" in photoelectric emission. The threshod frequency of a metal is f. When the light of frequency 2f is incident on the metal plate, the maximum velocity of photo-electrons is v1. When the frequency of the incident radiation is increased to 5f, the maximum velocity of phto-electrons is v2. Find the ratio v1 : v2.
Visible light has wavelengths in the range of 400 nm to 780 nm. Calculate the range of energy of the photons of visible light.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Plot a graph to show the variation of stopping potential with frequency of incident radiation in relation to photoelectric effect.
Work function of aluminium is 4.2 eV. If two photons each of energy 2.5 eV are incident on its surface, will the emission of electrons take place? Justify your answer.
In the experimental set up for studying photoelectric effect, if keeping the frequency of the incident radiation and the accelerating potential fixed, the intensity of light is varied, then ______.
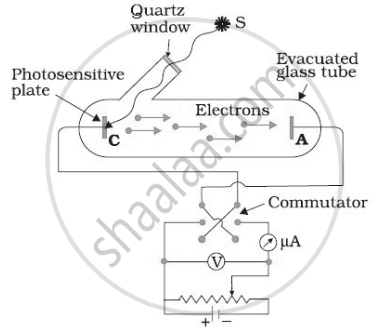
For a given frequency of light and a positive plate potential in the set up below, If the intensity of light is increased then ______.
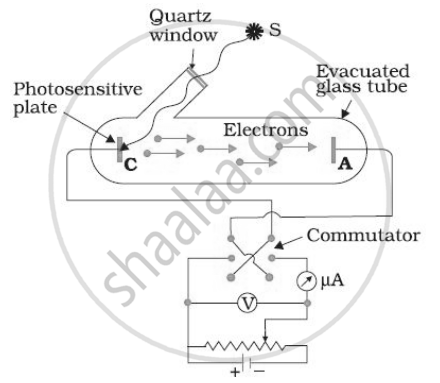
In various experiments on photo electricity, the stopping potential for a given frequency of the incident radiation is ______.
The electromagnetic theory of light failed to explain ______.
An increase in the intensity of the radiation causing photo-electric emission from a surface does not affect the maximum K.E. of the photoelectrons. Explain.
