Advertisements
Advertisements
प्रश्न
What are cathode rays? How are these rays formed?
उत्तर
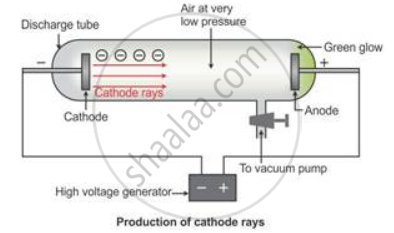
Cathode rays are the beam of electrons which travel from the negatively charged end (i.e. cathode to anode) of a vacuum tube, across a voltage difference between the electrodes placed at each end.
Formation of cathode rays:
Generally gases are poor conductors of electricity. However, when a high voltage charge from an induction coil is applied to tubes filled with gases at very low pressure (0.01 mm of mercury), the gases become good conductors of electricity and begin to flow in the form of rays.
These rays are called cathode rays and they travel from the cathode towards the anode.
APPEARS IN
संबंधित प्रश्न
Name the sub-atomic particle whose charge is -1.
Write down the number of electrons in X and neutrons in Y.
The electronic configuration of fluoride ion is the same as that of the neon atom. What is the difference between the two?
Name or state the following:
A bond formed by transfer of electron(s)
What is the relation between the number of protons and the number of electrons in an atom?
Differentiate between the following term:
Electron and proton
Atoms of all metals will have ______ electrons in their outermost orbit.
The maximum number of electrons that can be present in the N shell is ______.
When an atom gains an electron it has more electrons and thus it carries a positive charge.
Select the INCORRECT statement.
