Advertisements
Advertisements
प्रश्न
What are inner transition elements?
उत्तर
Lanthanoids and actinoids are called inner transition elements because inner f-orbitals are progressively filled and the last electron goes to the anti-penultimate f-orbital.
APPEARS IN
संबंधित प्रश्न
Why do interstitial compounds have higher melting points than corresponding pure metals?
Which metal in the first transition series (3d series) exhibits + 1 oxidation state most frequently and why?
Account for the following:
Cr2+ is a strong reducing agent.
The elements of 3d transition series are given as: Sc Ti V Cr Mn Fe Co
Answer the following: Which element has the highest m.p?
Give reasons: Transition metals show variable oxidation states.
How would you account for the irregular variation of ionization enthalpies (first and second) in the first series of the transition elements?
Which of the d-block elements may not be regarded as the transition elements?
Compare the stability of +2 oxidation state for the elements of the first transition series.
What are alloys?
NF3 is possible, but NF5 is not. Why?
How would you account for the following?
Zr (Z = 40) and Hf (Z = 72) have almost identical radii.
Give reasons Iron has the higher enthalpy of atomization than that of copper.
Which of the following ions show higher spin only magnetic moment value?
(i) \[\ce{Ti^3+}\]
(ii) \[\ce{Mn2+}\]
(iii) \[\ce{Fe2+}\]
(iv) \[\ce{Co3+}\]
When a brown compound of manganese (A) is treated with \[\ce{HCl}\] it gives a gas (B). The gas taken in excess, reacts with \[\ce{NH3}\] to give an explosive compound (C). Identify compounds A, B and C.
Reactivity of transition elements decreases almost regularly from Sc to Cu. Explain.
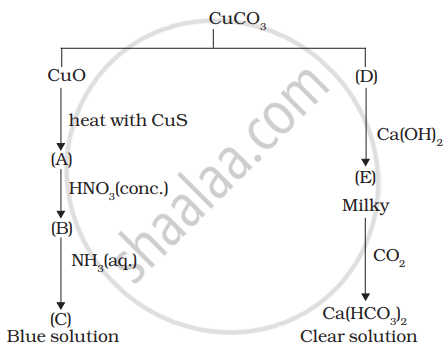
Identify A to E and also explain the reactions involved.
The orientation of an atomic orbital is governed by
Which of the following maxm magnetic moment?
The value of Δ0 for \[\ce{RhCl^{3-}6}\] is 243 KJ/mol which wavelength of light will promote an electron from. The colour of the complex is ______.
