Advertisements
Advertisements
प्रश्न
What is "baking powder"? How does it make the cake soft and spongy?
उत्तर
Baking powder is a sodium salt called sodium bicarbonate (NaHCO3). It is a mixture of baking soda (sodium hydrogencarbonate) and a mild edible acid such as tartaric acid. When baking soda mixes with water, the sodium hydrogencarbonate reacts with the tartaric acid to produce carbon dioxide gas. The carbon dioxide gas produced gets trapped in the wet dough made for baking a cake or bread, and bubbles out slowly, and the cake or bread becomes soft and spongy.
APPEARS IN
संबंधित प्रश्न
What is the common name of the compound CaOCl2?
Which compound of calcium is used for disinfecting drinking water supply?
Which is the real bleaching agent present in bleaching powder?
What happens when bleaching powder reacts with dilute sulphuric acid? Give equation of the reaction involved.
State two important uses of bleaching powder.
A white powdery substance having strong smell of chlorine is used for disinfecting drinking water supply at waterworks. Identify the substance. Give its chemical name and write the chemical reaction for its preparation.
When chlorine and sodium hydroxide being produced during the electrolysis of brine are allowed to mix, a new chemical is formed. Name the chemical and write its uses.
CaOCl2 will liberate Cl2 gas in the presence of
(i) CO2
(ii) HCl
(iii) CO
(iv) NO
The reaction between MnO2 with HCl is depicted in the following diagram. It was observed that gas with bleaching abilities was released.
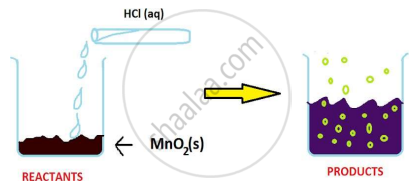
The chemical reaction between MnO2 and HCl is an example of:
Chlorine gas reacts with ____________ to form bleaching powder.
