Advertisements
Advertisements
प्रश्न
What do you observe when Ammonia solution is added drop by drop and then in excess to aqueous copper sulphate solution.
उत्तर
Initially when ammonium solution is added to aqueous solution of
copper sulphate a bluish precipi tate of copper hydroxide is obtained
which dissolves in excess of ammonium hydroxide.
\[\ce{CuSO4 + 2NH4OH -> (NH4)2SO4 + Cu(OH)2}\]
\[\ce{Cu(OH)2 + (NH4)2SO4 + 2NH4OH -> (Cu(NH3)4)SO4 + 4H2O}\]
APPEARS IN
संबंधित प्रश्न
List the properties of ammonia that make it
(i) A good refrigerant
(ii) A cleaning agent
(iii) As a source of hydrogen
Choose the correct word or phrase from the bracket to complete the following sentence:
Heating ammonium chloride with sodium hydroxide produces ______.
The diagram shows a simple arrangement of the fountain experiment:
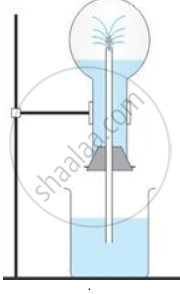
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
Name the gas evolved when the following mixtures are heated:
Sodium nitrate and ammonium chloride.
State one relevant observation of burning of ammonia in air.
Given one test can be used to detect the presence of the ion produced
What do you observe when Ammonia in excess is mixed with chlorine.
Identify the terms for the following:
The experiment which demonstrates high solubility of ammonia gas.
