Advertisements
Advertisements
प्रश्न
The diagram shows a simple arrangement of the fountain experiment:
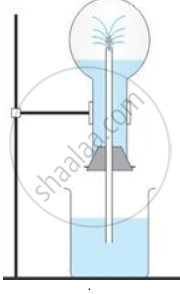
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
उत्तर
(i) Ammonia and hydrogen chloride gas
(ii) High solubility of gases in water
APPEARS IN
संबंधित प्रश्न
Complete the following equation. What property of ammonia is illustrated by the reaction given below.
2NH3 + 3CuO →
Identify the substance underlined, in the following case:
Cation that does not form a precipitate with ammonium hydroxide but forms one with sodium hydroxide.
State one relevant observation of burning of ammonia in air.
What do you observe when Ammonia solution is added drop by drop and then in excess to aqueous copper sulphate solution.
Differentiate between:
Aqueous ferrous and ferric sulphate solution
Write an equation to illustrate the reducing nature of ammonia.
State a relevant reason for the following:
Ammonia gas is not collected over water.
