Advertisements
Advertisements
प्रश्न
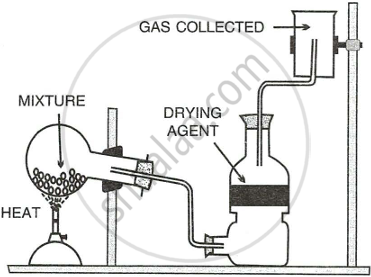
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.

- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
The diagram below shows the set up for the laboratory preparation of a pungent alkaline gas.
- Name the gas collected in the jar.
- Give a balanced equation for the above preparation.
- State how the above gas is collected.
- Name the drying agent used.
- State how you will find out that the jar is full of the pungent gas.
उत्तर
- Ammonia
- \[\ce{NH4CI + Ca(OH)2 ->[Heat] CaCI2 + 2H2O + 2NH3↑}\]
- By downward displacement of air
- Quicklime \[\ce{(CaO)}\]
- Bring moist red litmus paper to the mouth of the inverted jar; it immediately turns blue.
Or
Bring a glass rod dipped in hydrochloric acid to the mouth of the inverted jar. If it produces dense white fumes, then the jar is full of gas.
APPEARS IN
संबंधित प्रश्न
Ammonium salts decompose on heating. What other property do ammonium salts have in common?
Name a metallic chloride soluble in ammonium hydroxide.
Describe two tests to identify ammonia and ammonium ions in an aqueous solution.
Identify the substance underlined, in the following case:
A solid formed by reaction of two gaes, one of which is acidic and the other basic in nature.
What do you observe when Ammonia in excess is mixed with chlorine.
What do you observe when Filter paper dipped in colourless phenolphthalein is introduced into ammonia.
What do you observe when Ammonia comes in contact with the eyes of a person.
Choose the correct answer from the options given below :
Ammonia is produced when ammonium chloride is heated with
How is ammonia soluble in water?
State a relevant reason for the following:
Ammonia gas is not collected over water.
