Advertisements
Advertisements
Questions
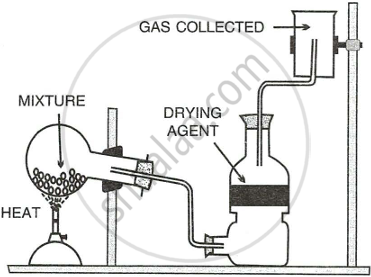
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.

- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
The diagram below shows the set up for the laboratory preparation of a pungent alkaline gas.
- Name the gas collected in the jar.
- Give a balanced equation for the above preparation.
- State how the above gas is collected.
- Name the drying agent used.
- State how you will find out that the jar is full of the pungent gas.
Solution
- Ammonia
- \[\ce{NH4CI + Ca(OH)2 ->[Heat] CaCI2 + 2H2O + 2NH3↑}\]
- By downward displacement of air
- Quicklime \[\ce{(CaO)}\]
- Bring moist red litmus paper to the mouth of the inverted jar; it immediately turns blue.
Or
Bring a glass rod dipped in hydrochloric acid to the mouth of the inverted jar. If it produces dense white fumes, then the jar is full of gas.
APPEARS IN
RELATED QUESTIONS
What property of ammonia is demonstrated by the fountain experiment?
State what you observe when a piece of moist red litmus paper is placed in a gas jar of ammonia.
Describe two tests to identify ammonia and ammonium ions in an aqueous solution.
Choose the correct word or phrase from the bracket to complete the following sentence:
Heating ammonium chloride with sodium hydroxide produces ______.
What do you observe when Ammonium solution is added to ferric chloride solution.
What do you observe when Ammonia solution is added drop by drop and then in excess to aqueous copper sulphate solution.
What do you observe when Ammonia comes in contact with the eyes of a person.
Differentiate between:
Action on indicators or dry ammonia gas and aqueous ammonia
How is ammonia soluble in water?
