Advertisements
Advertisements
Question
How is ammonia soluble in water?
Solution
When ammonia dissolve in water, it form ammonium hydroxide which
dissociates into NH4+ and OH-ion . Therefore it become soluble in water.
APPEARS IN
RELATED QUESTIONS
State what you observe when a piece of moist red litmus paper is placed in a gas jar of ammonia.
Complete the following equation. What property of ammonia is illustrated by the reaction given below.
2NH3 + 3CuO →
What is liquor ammonia fortis?
How will you demonstrate the solubility of ammonia in water? Explain.
List the properties of ammonia that make it
(i) A good refrigerant
(ii) A cleaning agent
(iii) As a source of hydrogen
The diagram shows a simple arrangement of the fountain experiment:
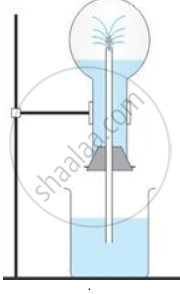
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
Name the other ion formed when ammonia dissolves in water
What do you observe when Ammonia in excess is mixed with chlorine.
What do you observe when Filter paper dipped in colourless phenolphthalein is introduced into ammonia.
What do you observe when Ammonia comes in contact with the eyes of a person.
