Advertisements
Advertisements
प्रश्न
What do you observe when when dilute sulphuric acid is added to barium chloride solution ?
उत्तर
When few drops of dilute sulphuric acid is added to barium chloride solution, a white precipitate of barium sulphate is formed.
\[\ce{\underset{\text{Barium chloride solution}}{BaCl2}+\underset{\text{Sulphuric acid}}{H2SO4}->\underset{\text{(Barium sulphate)(White precipitate)}}{BaSO4 ↓} +\underset{\text{Hydrochloride acid}}{2HCl}}\]
APPEARS IN
संबंधित प्रश्न
What type of reaction is represented by the following equation?
2 Ca + O2 → 2CaO
Give one example of a combination reaction which is also exothermic.
Give one example of a combination reaction which is also endothermic.
When hydrogen burns in oxygen, water is formed and when water is electrolysed, then hydrogen and oxygen are produced. What type of reaction takes place:
(a) in the first case?
(b) in the second case
What do you understand by a chemical bond ?
Fill in the blank
The chemical reaction between hydrogen and chlorine is a ................ reaction
Classify the following reaction into –
- Direct combination
- Decomposition
- Displacement
- Double decomposition
The reaction is – Molten zinc at high temperatures, burns in air to give zinc oxide.
Give a balanced equation for the following type of reaction:
A reaction of direct combination i.e. synthesis in which twp gases combine to give another gas – which turns lime water milky.
The Salt Story
From: The New Indian Express 9 March 2021
The salt pans in Marakkanam, a port town about 120 km from Chennai are the third largest producer of salt in Tamil Nadu. Separation of salt from water is a laborious process and the salt obtained is used as raw materials for manufacture of various sodium compounds.
One such compound is Sodium hydrogen carbonate, used in baking, as an antacid and in soda acid fire extinguishers.
The table shows the mass of various compounds obtained when 1litre of sea water is evaporated.
| COMPOUND | FORMULA | MASS OF SOLID PRESENT /g |
| Sodium Chloride | NaCl | 28.0 |
| Magnesium Chloride | MgCl2 | 8.0 |
| Magnesium Sulphate | MgSO4 | 6.0 |
| Calcium Sulphate | CaSO4 | 2.0 |
| Calcium Carbonate | CaCO3 | 1.0 |
| TOTAL AMOUNT OF SALT OBTAINED | 45.0 | |
Which compound in the table reacts with acids to release carbon dioxide?
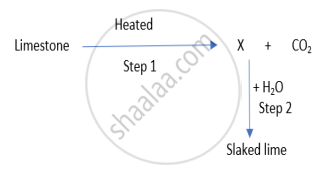
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
