Advertisements
Advertisements
प्रश्न
What do you observe when when dilute sulphuric acid is added to barium chloride solution ?
उत्तर
When few drops of dilute sulphuric acid is added to barium chloride solution, a white precipitate of barium sulphate is formed.
\[\ce{\underset{\text{Barium chloride solution}}{BaCl2}+\underset{\text{Sulphuric acid}}{H2SO4}->\underset{\text{(Barium sulphate)(White precipitate)}}{BaSO4 ↓} +\underset{\text{Hydrochloride acid}}{2HCl}}\]
APPEARS IN
संबंधित प्रश्न
What type of reaction is represented by the following equation?
CaO + H2O → Ca(OH)
What type of chemical reaction take place when ammonia and hydrogen chloride are mixed?
Give one example of a combination reaction which is also endothermic.
What happens during a chemical reaction ?
What do you observe when solid lead nitrate is heated?
Calcium oxide reacts vigorously with water to produce slaked lime.
\[\ce{CaO{(s)} + H2O(l) -> Ca(OH)2(aq)}\]
This reaction can be classified as:
(A) Combination reaction
(B) Exothermic reaction
(C) Endothermic reaction
(D) Oxidation reaction
Define a combination reaction. Give one example of an exothermic combination reaction.
Combustion reactions are always ____________.
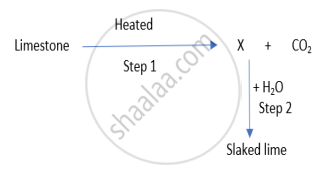
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
Read the text below and answer the questions that follow:
A small amount of hydrochloric acid was taken in a test tube. The test tube was heated. A glass rod was dipped in the ammonia solution and held on the top of the test tube. A white smoke was seen emanating from the tip of the glass rod.
- What must have happened?
- Which colour of gas is formed?
- Write the chemical equation for the reaction.
