Advertisements
Advertisements
प्रश्न
What happens during a chemical reaction ?
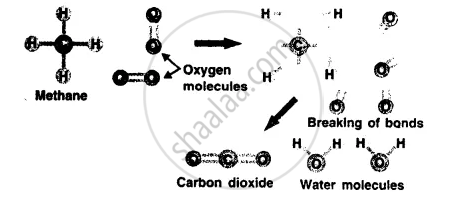
उत्तर
A chemical reaction involves breaking of chemical bonds between the atoms or groups of atoms of reacting substances and rearrangement of atoms making new bonds to form new substances.
APPEARS IN
संबंधित प्रश्न
What type of reaction is represented by the following equation?
CaO + H2O → Ca(OH)
What type of chemical reaction take place when a magnesium wire is burnt in air?
When hydrogen burns in oxygen, water is formed and when water is electrolysed, then hydrogen and oxygen are produced. What type of reaction takes place:
(a) in the first case?
(b) in the second case
Fill in the blank
A reaction in which two or more substances combine to form a single substance is called a ............ reaction.
Classify the following reaction as combination, decomposition, displacement, precipitation and neutralization. Also balance the equation.
\[\ce{CaCO3_{(s)} ->[heat]CaO_{(s)} + CO2_{(g)}}\]
Explain the terms with examples.
Combination reaction
Define: Chemical combination reaction
Give a balanced equation for –
A direct combination reaction involving two elements, one of which is a non-metal
The respiration process during which glucose undergoes slow combustion by combining with oxygen in the cells of our body to produce energy is a kind of:
Balance the following chemical equation and identify the type of chemical reaction.
`"Na"("s") + "S"("s") overset("(Fuse)")(->) "Na"_2"S"("s")`
