Advertisements
Advertisements
Question
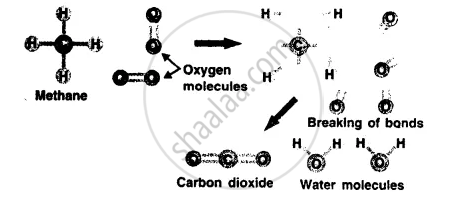
What happens during a chemical reaction ?
Solution
A chemical reaction involves breaking of chemical bonds between the atoms or groups of atoms of reacting substances and rearrangement of atoms making new bonds to form new substances.
APPEARS IN
RELATED QUESTIONS
What do you understand by a chemical bond ?
What do you observe when solid lead nitrate is heated?
What do you observe when when dilute sulphuric acid is added to barium chloride solution ?
Fill in the blank
A reaction in which two or more substances combine to form a single substance is called a ............ reaction.
(a) What is double displacement reaction? Explain with an example.
(b) A small amount of quick lime is added to water in a beaker.
(i) Name and define the type of reaction that has taken place.
(ii) Write balanced chemical equation for the above reaction and the chemical name of the product formed.
(iii) List two main observations of this reaction.
The respiration process during which glucose undergoes slow combustion by combining with oxygen in the cells of our body to produce energy is a kind of:
Identify the types of chemical reactions occurring during the combustion of fuel:
Balance the following chemical equation and identify the type of chemical reaction.
`"Mg"("s") + "Cl"_2("g") -> "MgCl"_2("s")`
Give the chemical reaction when a few pieces of slaked lime are added to a beaker half-filled with water. Will it be an endothermic or exothermic reaction?
- Assertion: Rusting of Iron is endothermic in nature.
- Reason: As the reaction is slow, the release of heat is barely evident.
