Advertisements
Advertisements
प्रश्न
What happens chemically when quicklime is added to water filled in a bucket?
उत्तर
When quicklime (calcium oxide) is added to water filled in a bucket, they react chemically to form slaked lime (calcium hydroxide) and produce heat energy. So, water in the bucket becomes hot.

APPEARS IN
संबंधित प्रश्न
Find the odd man out:
Camphor, Ammonium Chloride, Naphthalene balls, Sugar
What type of reaction is represented by the following equation?
CaO + H2O → Ca(OH)
Define a combination reaction.
What do you understand by a chemical bond ?
Explain the following type of chemical reaction, giving two examples for it:
Combination reaction
Give a balanced equation for the following type of reaction:
A reaction of direct combination i.e. synthesis in which twp gases combine to give another gas – which turns lime water milky.
Which changes occur during chemical changes?
Which of the following is not a physical change?
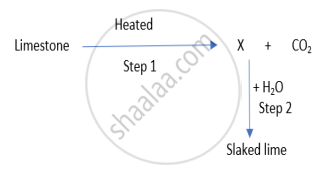
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
A clear solution of slaked lime is made by dissolving Ca(OH)2 in an excess of water. This solution is left exposed to air. The solution slowly goes milky as a faint white precipitate forms. Explain why a faint white precipitate forms, support your response with the help of a chemical equation.
