Advertisements
Advertisements
Question
What happens chemically when quicklime is added to water filled in a bucket?
Solution
When quicklime (calcium oxide) is added to water filled in a bucket, they react chemically to form slaked lime (calcium hydroxide) and produce heat energy. So, water in the bucket becomes hot.

APPEARS IN
RELATED QUESTIONS
One of the following is an endothermic reaction. This is:
(a) combination of carbon and oxygen to form carbon monoxide
(b) combination of nitrogen and oxygen to form nitrogen monoxide
(c) combination of glucose and oxygen to form carbon dioxide and water
(d) combination of zinc and hydrochloric acid to form zinc chloride and hydrogen
What type of reaction is represented by the following equation?
CaO + H2O → Ca(OH)
Define a combination reaction.
What happens during a chemical reaction ?
What do you understand by a chemical bond ?
Explain the following type of chemical reaction, giving two examples for it:
Combination reaction
The number of products formed in a synthesis reaction is ______
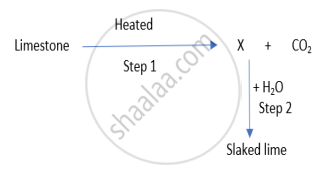
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
A metal ribbon 'X' bums in oxygen with a dazzling white flame forming a white ash 'Y'. The correct description of X, Y and the type of reaction is:
- Assertion: Rusting of Iron is endothermic in nature.
- Reason: As the reaction is slow, the release of heat is barely evident.
