Advertisements
Advertisements
Question
The number of products formed in a synthesis reaction is ______
Solution
The number of products formed in a synthesis reaction is One.
APPEARS IN
RELATED QUESTIONS
One of the following is an endothermic reaction. This is:
(a) combination of carbon and oxygen to form carbon monoxide
(b) combination of nitrogen and oxygen to form nitrogen monoxide
(c) combination of glucose and oxygen to form carbon dioxide and water
(d) combination of zinc and hydrochloric acid to form zinc chloride and hydrogen
What type of reaction is represented by the following equation?
CaO + H2O → Ca(OH)
what tpye of reaction is the following:
2K + Cl → 2KCl
When hydrogen burns in oxygen, water is formed and when water is electrolysed, then hydrogen and oxygen are produced. What type of reaction takes place:
(a) in the first case?
(b) in the second case
Fill in the blank
The chemical reaction between hydrogen and chlorine is a ................ reaction
What effect does an increase in concentration of H+ (aq.) in a solution have on the pH of solution?
(a) What is double displacement reaction? Explain with an example.
(b) A small amount of quick lime is added to water in a beaker.
(i) Name and define the type of reaction that has taken place.
(ii) Write balanced chemical equation for the above reaction and the chemical name of the product formed.
(iii) List two main observations of this reaction.
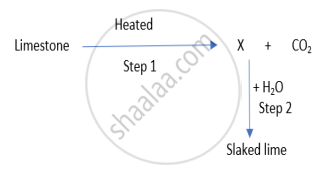
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
Identify the types of chemical reactions occurring during the combustion of fuel:
A clear solution of slaked lime is made by dissolving Ca(OH)2 in an excess of water. This solution is left exposed to air. The solution slowly goes milky as a faint white precipitate forms. Explain why a faint white precipitate forms, support your response with the help of a chemical equation.
