Advertisements
Advertisements
Question
what tpye of reaction is the following:
2K + Cl → 2KCl
Solution
Combination reaction
APPEARS IN
RELATED QUESTIONS
A silvery-white metal X taken in the form of ribbon, when ignited, burns in air with a dazzling white flame to form a white powder Y. When water is added to powder Y, it dissolves partially to form another substance Z.
(a) What could metal X be?
(b) What is powder Y?
(c) With which substance metal X combines to form powder Y?
(d) What is substance Z? Name one domestic use of substance Z.
(e) Write a balanced chemical equation of the reaction which takes place when metal X burns in air to form powder Y.
What type of reaction is represented by the following equation?
CaO + CO2 → CaCO3
A student prepared an aqueous solution of CuSO4 in beaker X and an aqueous solution of FeSO4 in beaker Y. He then dropped some iron pieces in beaker X and some zinc pieces in beaker Y. After about 10 hours he observed that the solutions in X and Y respectively appear:
(A) blue and green
(B) colourless and pale green
(C) colourless and light blue
(D) greenish and colourless
Select the correct answer for the statement given below:
The product formed during direct combination reaction of carbon dioxide and water.
Give a balanced equation for the following type of reaction:
A reaction of direct combination i.e. synthesis in which twp gases combine to give another gas – which turns lime water milky.
Define a combination reaction. Give one example of an exothermic combination reaction.
Which of the following is not a physical change?
The respiration process during which glucose undergoes slow combustion by combining with oxygen in the cells of our body to produce energy is a kind of:
The chemical reaction between copper and oxygen can be categorized as ______.
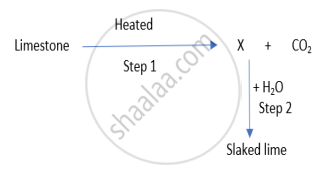
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
