Advertisements
Advertisements
Question
A metal ribbon 'X' bums in oxygen with a dazzling white flame forming a white ash 'Y'. The correct description of X, Y and the type of reaction is:
Options
X = Ca; Y = CaO;
Type of reaction = Decomposition
X = Mg; Y = MgO;
Type of reaction = Combination
X = Al; Y = Al2O3;
Type of reaction = Thermal decomposition
X = Zn; Y = ZnO;
Type of reaction = Endothermic
Solution
X = Mg; Y = MgO;
Type of reaction = Combination
Explanation:
In this reaction, magnesium and oxygen combine to create magnesium oxide.
\[\ce{2Mg + O_2 ->2MgO}\]
APPEARS IN
RELATED QUESTIONS
What happens chemically when quicklime is added to water filled in a bucket?
What type of reaction is represented by the following equation?
CaO + H2O → Ca(OH)
What type of chemical reaction take place when ammonia and hydrogen chloride are mixed?
What type of chemical reaction is represented by the following equation?
A + B → C
What do you understand by a chemical bond ?
What effect does an increase in concentration of H+ (aq.) in a solution have on the pH of solution?
Explain the following reaction with their balanced chemical equation.
Ammonia gas reacts with hydrogen chloride.
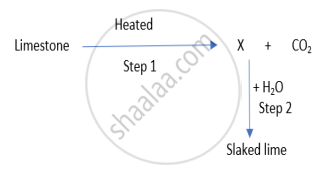
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
Give the chemical reaction when a few pieces of slaked lime are added to a beaker half-filled with water. Will it be an endothermic or exothermic reaction?
Read the text below and answer the questions that follow:
A small amount of hydrochloric acid was taken in a test tube. The test tube was heated. A glass rod was dipped in the ammonia solution and held on the top of the test tube. A white smoke was seen emanating from the tip of the glass rod.
- What must have happened?
- Which colour of gas is formed?
- Write the chemical equation for the reaction.
