Advertisements
Advertisements
प्रश्न
What happens when oxidation of tert-butylamine with KMnO4?
उत्तर
In general, primary amines, in which the –NH2 group is attached to a tertiary carbon can be oxidised with KMnO4 to the corresponding nitro compound in excellent yield.
Therefore 3°-butylamine oxidised to give 2-methyl-2-nitropropane.
\[\begin{array}{cc}
\phantom{...}\ce{CH3}\phantom{...................}\ce{CH3}\phantom{}\\
\phantom{}|\phantom{.......................}|\phantom{}\\
\ce{CH3 - C - NH2 ->[KMnO4] CH3 - C - NO2}\\
\phantom{}|\phantom{.......................}|\phantom{}\\
\phantom{........}\ce{\underset{(3°-butylamine)}{CH3}}\phantom{........}\ce{\underset{(2-methyl-2-nitropropane)}{CH3}}\phantom{..}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Which of the following reagent can be used to convert nitrobenzene to aniline?
Which one of the following nitro compounds does not react with nitrous acid?
Which of the following reaction is not correct.
Nitrobenzene on reaction with at 80-100°C forms which one of the following products?
Among the following, the reaction that proceeds through an electrophilic substitution is:
How will you convert nitrobenzene into azoxybenzene?
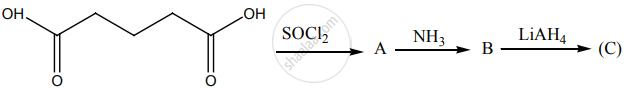
Identify A, B and C.
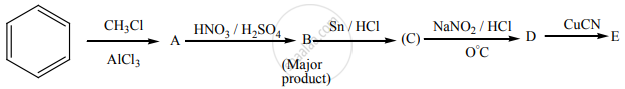
Identify A to E in the following sequence of reactions.
Account for the following.
Aniline does not undergo Friedel – Crafts reaction
Account for the following.
Aniline does not undergo Friedel–Crafts reaction.
