Advertisements
Advertisements
प्रश्न
What is the difference between:
Strong electrolyte and weak electrolyte.
उत्तर
| Strong Electrolytes | Weak Electrolytes | |
| (1) | Electrolytes which allow a large amount of electricity to flow through them. | Electrolytes which allow small amounts of electricity to flow through them. |
| (2) | These are good conductors of electricity. | These are poor conductors of electricity. |
| (3) | These are almost completely dissociated in the fused or aqueous solution state. | These are partially dissociated in the fused or aqueous solution state. |
| (4) | These solutions contain (almost) only free mobile ions. | These solutions contain ions as well as molecules. |
| (5) | Examples: | Examples: |
| Acids: Hydrogen chloride, Sulphuric acid, Nitric acid, etc. | Acids: Carbonic acid, acetic acid, oxalic acid, etc. | |
| Bases: NaOH, KOH. (aqueous or molten state) | Bases: Ammonium hydroxide, calcium hydroxide. (aqueous or molten state) | |
| Salts: NaCl (molten or aqueous), PbBr2 (molten), CuCl2 (aq.), CuSO4 (aq.) | Salts: Ammonium carbonate and lead acetate. |
APPEARS IN
संबंधित प्रश्न
Give appropriate scientific reasons for During electrolysis of molten lead bromide, graphite anode is preferred to other electrodes.
Name the gas that is produced in each of the given case :
At the anode during the electrolysis of acidified water.
Copy and complete the following table which refers to the conversion of ions to neutral particles.
| Conversion | Ionic equation | Oxidation / Reduction |
| Chloride ion to chlorine molecule | 1)_________ | 2)_________ |
| Lead (II) ion to lead | 3)_________ | 4)_________ |
Define the term : Electrolyte
What should be the physical state of lead bromide if it is to conduct electricity?
Correct the following statement:
Lead bromide conducts electricity.
Three different electrolytic cells, A, B and C are connected in separate circuits. Electrolytic cell A contains a sodium chloride solution. When the circuit is completed, a bulb in the circuit glows brightly. Electrolytic cell B contains an acetic acid solution and in this case, the bulb in the circuit glows dimly. The electrolytic cell C contains a sugar solution and the bulb does not glow. Give a reason for each of these observations.
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
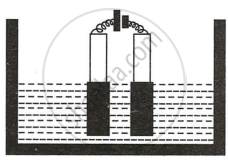
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Identify the substance underlined in each of the following case :
The particles present in a liquid such as kerosene, that is non-electrolyte.
Classify the following substance:
Dilute sulphuric acid
