Advertisements
Online Mock Tests
Chapters
2: Chemical Bonding
3: Acids, Bases and Salts
4: Analytical Chemistry
5: Mole concept and Stoichiometry
▶ 6: Electrolysis
7: Metallurgy
8: Study of Compounds A - Hydrogen Chloride
9: Study of Compounds B - Ammonia
10: Study of Compounds C - Nitric Acid
11: Study of Compounds D - Sulphuric Acid
12: Organic Chemistry
13: Practical Work
![Selina solutions for Concise Chemistry [English] Class 10 ICSE chapter 6 - Electrolysis Selina solutions for Concise Chemistry [English] Class 10 ICSE chapter 6 - Electrolysis - Shaalaa.com](/images/concise-chemistry-english-class-10-icse_6:a11ba386c5de4fb5831d789b303b585d.jpg)
Advertisements
Solutions for Chapter 6: Electrolysis
Below listed, you can find solutions for Chapter 6 of CISCE Selina for Concise Chemistry [English] Class 10 ICSE.
Selina solutions for Concise Chemistry [English] Class 10 ICSE 6 Electrolysis lntext Questions [Pages 107 - 113]
Fill in the blanks.
Powdered sodium chloride (common salt) does not conduct an electric current, but it does so when ______ or when ______.
Molten lead bromide conducts electricity. It is called an ______. It is composed of lead ______ and bromide ______. The lead ions are ______ charged and are called ______. The bromide ______ are ______ charged and are called ______.
Substances which conduct electricity in the solid state are generally ______.
The electron releasing tendency of zinc is ______ than that of copper.
A solution of HCl gas in water conducts electricity because ______, but a solution of HCl gas in toluene does not conduct an electric current because ______.
Pure water consists entirely of ______.
ions
molecules
We can expect that pure water ______ normally conducts electricity.
will
will not
Electrolysis is the passage of ______ through a liquid or a solution accompanied by a ______ change.
electricity
electrons
physical
chemical
Define the following term:
Electrolysis
Define the following term:
Non-electrolyte
Define the following term:
Cation
Define the following term:
anion
Define the following term:
Weak electrolytes
What is the difference between:
Modern explanation and Arrhenius explanation for the theory of electrolysis.
What is the difference between:
Electrolytic dissociation and ionisation.
What is the difference between:
A cation and an anion
What is the difference between:
Electrolytic dissociation and thermal dissociation.
What is the difference between:
Strong electrolyte and weak electrolyte.
Name a salt which is a weak electrolyte.
Name a base which is a weak electrolyte.
Name an inert electrode.
Name an active electrode.
Name a positively charged non-metallic ion.
Name the electrode at which reduction occurs.
Name a non-metallic element which is a conductor of electricity.
Electrolysis is a redox process. Explain.
Classify the following substance:
Acetic acid
Strong electrolyte
Weak electrolyte
Non-electrolyte
Classify the following substance:
Ammonium chloride
Strong electrolyte
Weak electrolyte
Non-electrolyte
Classify the following substance:
Ammonium hydroxide
Strong electrolyte
Weak electrolyte
Non-electrolyte
Classify the following substance:
Carbon tetrachloride
Strong electrolyte
Weak electrolyte
Non-electrolyte
Classify the following substance:
Dilute hydrochloric acid
Strong electrolyte
Weak electrolyte
Non-electrolyte
Classify the following substance:
Sodium acetate
Strong electrolyte
Weak electrolyte
Non-electrolyte
Classify the following substance:
Dilute sulphuric acid
Strong electrolyte
Weak electrolyte
Non-electrolyte
Explain why Cu, though a good conductor of electricity is a non electrolyte?
Explain why solid sodium chloride does not allow electricity to pass through?
Choose the option to match the description given below. Some letters may be repeated.
Molten ionic compound
Non-electrolyte
Strong electrolyte
Weak electrolyte
Metallic conductor
Choose the option to match the description given below. Some letters may be repeated.
Carbon tetrachloride
Non-electrolyte
Strong electrolyte
Weak electrolyte
Metallic conductor
Choose the option to match the description given below. Some letters may be repeated.
An aluminium wire
Non-electrolyte
Strong electrolyte
Weak electrolyte
Metallic conductor
Choose the option to match the description given below. Some letters may be repeated.
A solution containing solvent molecules, solute molecules and ions formed by the dissociation of solute molecules.
Non-electrolyte
Strong electrolyte
Weak electrolyte
Metallic conductor
Choose the option to match the description given below. Some letters may be repeated.
A sugar solution with sugar molecules and water molecules.
Non-electrolyte
Strong electrolyte
Weak electrolyte
Metallic conductor
An electrolyte which completely dissociates into ions is ______.
Alcohol
Carbonic acid
Sucrose
Sodium hydroxide
Name two substances in the following case:
Contain only molecules.
Name two substances in the following case:
Contain only ions.
Name two substances in the following case:
Contain ions as well as molecules.
Select the ion in the following case, that would get selectively discharged from the aqueous mixture of the ions listed below:
\[\ce{SO^2-_4, NO^-_3 {and} OH-}\]
Select the ion in the following case, that would get selectively discharged from the aqueous mixture of the ions listed below:
Pb2+, Ag+ and Cu2+
Among Zn and Cu, which would occur more readily in nature as metal and which as an ion?
Why cannot we store AgNO3 solution in copper vessel?
Out of Cu and Ag, which is more active?
How would you change a metal like Cu into its ions?
How would you change Cu2+ ion to Cu?
A solution of caustic soda (NaOH) in water or when fused, conducts an electric current. what is the similarity in these two cases?
During electrolysis of an aqueous solution of sulphuric acid between platinum electrodes, two types of anions migrate towards the anode but only one of them is discharged:
- Name the two anions.
- Name the main product of the discharge of anion at the anode and write the anode reaction.
- Name the product at the cathode and write the reaction.
- Do you notice any change in colour. State why?
- Why this electrolysis, is considered as an example of catalysis?
Copper sulphate solution is electrolysed using a platinum cathode and carbon anode.
Study the diagram given alongside and answer the following questions:
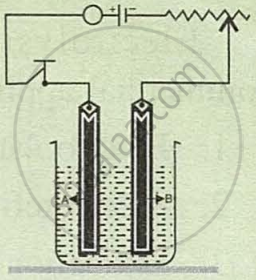
- Give the names of the electrodes A and B.
- Which electrode is the oxidising electrode?
To carry out the so-called "electrolysis of water", sulphuric acid is added to water. How does the addition of sulphuric acid produce a conducting solution?
Choosing only words from the following list, write down the appropriate words to fill in the blanks (a) to (e) below:
anions, anode, cathode, cations, electrode, electrolyte, nickel, voltameter.
The electroplating of an article with nickel requires an (a) ______ which must be a solution containing (b) ______ ions. The article to be plated is placed as the (c) ______ of the cell in which the plating is carried out. The (d) ______ of the cell is made from pure nickel. The ions that are attracted to the negative electrode and discharged are called (e) ______.
When a molten ionic compound is electrolysed, the metal is always formed at ______ and the non-metal is formed at ______.
Electrolysis of acidulated water is an example of ______.
Reduction
Oxidation
Redox reaction
Synthesis
Explain the following:
A solution of cane sugar does not conduct electricity, but a solution of sodium chloride is a good conductor.
Explain the following:
Hydrochloric acid is a good conductor of electricity.
Explain the following:
During the electrolysis of an aqueous solution of NaCI, hydrogen ion is reduced at the cathode and not the sodium ion though both Na+ and H+ ions are present in the solution.
Explain the following:
On electrolysis of dilute copper (II) sulphate solution, copper is deposited at the cathode but no hydrogen gas evolves there. Explain why?
Explain the following:
When a dilute aqueous solution of sodium chloride is electrolysed between platinum electrodes, hydrogen gas is evolved at the cathode but metallic sodium is not deposited. Why?
Explain the following:
Zinc can produce hydrogen on reacting with acids but copper cannot.
Selina solutions for Concise Chemistry [English] Class 10 ICSE 6 Electrolysis EXERCISE-6 [Pages 117 - 119]
Give reason for the following:
Electrolysis of molten lead bromide is considered to be a reaction in which oxidation and reduction go side by side, i.e., a redox reaction.
Give reason for the following:
The blue colour of aqueous copper sulphate fades when it is electrolysed using platinum electrodes.
Give reason for the following:
Lead bromide undergoes electrolytic dissociation in the molten state but is a non-electrolyte in the solid state.
Give reason for the following:
Aluminium is extracted from its oxide by electrolytic reduction and not by convectional reducing agents.
Give reason for the following:
The ratio of hydrogen and oxygen formed at the cathode and anode is 2 : 1 by volume.
Give reason for the following:
In the electrolysis of acidified water, dilute sulphuric acid is preferred to dilute nitric acid for acidification.
Give reason for the following:
Ammonia is unionised in the gaseous state but in the aqueous solution, it is a weak electrolyte.
Give reason for the following:
A graphite anode is preferred to other inert electrodes during the electrolysis of fused lead bromide.
Give reason for the following:
For electroplating with silver, silver nitrate is not used as an electrolyte.
Give reason for the following:
Carbon tetrachloride is a liquid but does not conduct electricity.
Give reason for the following:
Potassium is not extracted by electrolysis of its aqueous salt solution.
(a) Copy and complete the following table, which refers to two practical applications of electrolysis.
| Anode | Electrolyte | Cathode | |
| (i) Silver plating of a spoon | A solution of potassium argentocyanide | ||
| (ii) Purification of copper | |||
| (iii) Extraction of sodium |
(b) Write the equation taking place at the anode for Q. (a).
Draw a labelled diagram to show how iron is electroplated with copper.
Which solution is preferred as electrolyte, CuSO4 or FeSO4?
Describe what happens to the iron object and the copper rod.
Element X is a metal with a valency 2. Element Y is a non-metal with a valency 3.
Write equations to show how X and Y form ions?
Element X is a metal with a valency 2. Element Y is a non-metal with a valency 3.
If Y is a diatomic gas, write the equation for the direct combination of X and Y to form a compound.
Element X is a metal with a valency 2. Element Y is a non-metal with a valency 3.
If the compound formed between X and Y is melted and an electric current passed through the molten compound, the element X will be obtained at the ______ and Y at the ______ of the electrolytic cell. (Provide the missing words).
Write two applications of electrolysis in which the anode diminishes in mass.
What kind of particles will be found in a liquid compound which is a non-electrolyte?
If HX is a weak acid, what particles will be present in its dilute solution apart from those of water?
(Choose the correct word to fill in the blanks).
Cations are formed by ______ of electrons and anions are formed by ______ of electrons.
loss
gain
What ions must be present in a solution used for electroplating a particular metal?
A strip of copper is placed in four different colourless salt solutions. They are KNO3, AgNO3, Zn(NO3)2 and Ca(NO3)2. Which one of the solutions will finally turn blue?
Here is an electrode reaction:
\[\ce{Cu -> Cu^{2+} + 2e^-}\]
At which electrode (anode or cathode) would such a reaction take place? Is this an example of oxidation?
A solution contains magnesium ions (Mg+2), iron (II) ions (Fe+2) and copper ions (Cu+2). On passing an electric current through this solution, which ions will be the first to be discharged at the cathode? Write the equation for the cathode reaction.
Why is carbon tetrachloride, which is a liquid, a non-electrolyte?
During the electrolysis of molten lead bromide, which of the following takes place?
Bromine is released at the cathode.
Lead is deposited at the anode.
Bromine ions gain electrons.
Lead is deposited at the cathode.
Select the correct answer:
The aqueous solution of the compound which contains both ions and molecules is ______.
H2SO4
HCl
HNO3
CH3COOH
Correct the following statement:
Lead bromide conducts electricity.
A metal article is to be electroplated with silver. The electrolyte selected is sodium argentocyanide.
- What kind of salt is sodium argentocyanide?
- Why is it preferred to silver nitrate as an electrolyte?
- State one condition to ensure that the deposit is smooth, firm and long lasting.
- Write the reaction taking place at the cathode.
- Write the reaction taking place at the anode.
Aqueous solution of nickel sulphate contains \[\ce{Ni^{+2}}\] and \[\ce{SO^{-2}_{4}}\] ions.
- Which ion moves towards the cathode?
- What is the product at the anode?
Select the correct answer -
A compound which during electrolysis in its molten state, liberates a reddish brown gas at the anode.
Sodium chloride
Copper [II] oxide
Copper [II] sulphate
Lead [II] bromide
During electroplating of an article with nickel, name the electrolyte.
During electroplating of an article with nickel, name the cathode.
During electroplating of an article with nickel, name the anode.
During electroplating of an article with nickel, give the reaction of the electrolysis at the cathode.
During electroplating of an article with nickel, give the reaction of the electrolysis at the anode.
Three different electrolytic cells, A, B and C are connected in separate circuits. Electrolytic cell A contains a sodium chloride solution. When the circuit is completed, a bulb in the circuit glows brightly. Electrolytic cell B contains an acetic acid solution and in this case, the bulb in the circuit glows dimly. The electrolytic cell C contains a sugar solution and the bulb does not glow. Give a reason for each of these observations.
Give reasons as to why - the electrolysis of acidulated water is considered to be an example of catalysis.
During the electrolysis of copper (II) sulphate solution using platinum as cathode and carbon as anode:
- What do you observe at the cathode and at the anode?
- What change is noticed in the electrolyte?
- Write the reactions at the cathode and at the anode.
Differentiate between the electrical conductivity of copper sulphate solution and copper metal.
Identify the weak electrolyte from the following:
Sodium chloride solution
Dilute hydrochloric acid
Dilute sulphuric acid
Aqueous acetic acid
Match the following in column A with the correct answer from the choices given in column B:
| Column A | Column B |
| 1. Ammonium hydroxide solution | (i) Contains only ions |
| 2. Dilute hydrochloric acid | (ii) Contains only molecules |
| 3. Carbon tetrachloride | (iii) Contains ions and molecules |
Give reason:
An aqueous solution of sodium chloride conducts electricity.
Select the correct answer from the list in brackets:
An aqueous electrolyte consists of the ions mentioned in the list. The ion which could be discharged most readily during electrolysis.
\[\ce{Fe^{2+}}\]
\[\ce{Cu^{2+}}\]
\[\ce{H^+}\]
The metallic electrode which does not take part in an electrolytic reaction.
Cu
Ag
Pt
Ni
The ion which is discharged at the anode during the electrolysis of copper sulphate solution using platinum electrodes as anode and cathode.
\[\ce{Cu^{2+}}\]
\[\ce{OH^{-}}\]
\[\ce{SO^{2-}4}\]
\[\ce{H^+}\]
When dilute sodium chloride is electrolysed using graphite electrodes, the cation which is discharged at the cathode most readily.
\[\ce{Na^{1+}}\]
\[\ce{OH^{1-}}\]
\[\ce{H^{1+}}\]
\[\ce{Cl^{1-}}\]
During silver plating of an article using potassium argentocyanide as an electrolyte, the anode material should be.
Cu
Ag
Pt
Fe
State one observation when electricity is passed through molten lead bromide.
Which of these will act as a non-electrolyte?
Liquid carbon tetrachloride
Acetic acid
Sodium hydroxide aqueous solution acid
Potassium chloride aqueous solution
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
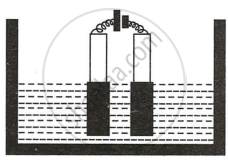
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Identify:
A gas which does not conduct electricity in the liquid state but conducts electricity when dissolved in water.
When fused lead bromide is electrolysed, we observe ______.
A silver grey deposit at the anode and a reddish brown deposit at the cathode.
A silver grey deposit at the cathode and a reddish brown deposit at the anode.
A silver grey deposit at the cathode and reddish brown fumes at the anode.
Silver grey fumes at the anode and reddish brown fumes at the cathode.
The electrolyte used for electroplating an article with silver is ______.
Silver nitrate solution
Silver cyanide solution
Sodium argentocyanide solution
Nickel sulphate solution
Give one word or phrase for the following:
Electrolytic deposition of a superior metal on a baser metal.
State your observation of the following case:
At the cathode when acidified aqueous copper sulphate solution is electrolyzed with copper electrodes.
Which electrode—anode or cathode—is the oxidising electrode? Why?
Name the kind of particles present in sodium hydroxide solution.
Name the kind of particles present in carbonic acid.
Name the kind of particles present in the sugar solution.
M is a metal above hydrogen in the activity series and its oxide has the formula M2O. The oxide when dissolved in water forms the corresponding hydroxide which is a good conductor of electricity. In the above context answer the following:
- What kind of combination exists between M and O?
- How many electrons are there in the outermost shell of M?
- Name the group to which M belongs.
- State the reaction taking place at the cathode.
- Name the product at the anode.
State one relevant observation for the following:
At the anode when aqueous copper sulphate solution is electrolysed using copper electrodes.
Give appropriate scientific reasons for the following statement:
During electrolysis of molten lead bromide, graphite anode is preferred to other electrodes.
Give reason for the following:
Electrolysis of molten lead bromide is considered to be a reaction in which oxidation and reduction go side by side, i.e., a redox reaction.
Give reasons why sodium chloride will conduct electricity only in the fused or aqueous solution state.
Give reasons why in the electroplating of an article with silver, the electrolyte sodium argentocynide solution is preferred over silver nitrate solution.
Give reasons why although copper is a good conductor of electricity, it is a non-electrolyte.
Solutions for 6: Electrolysis
![Selina solutions for Concise Chemistry [English] Class 10 ICSE chapter 6 - Electrolysis Selina solutions for Concise Chemistry [English] Class 10 ICSE chapter 6 - Electrolysis - Shaalaa.com](/images/concise-chemistry-english-class-10-icse_6:a11ba386c5de4fb5831d789b303b585d.jpg)
Selina solutions for Concise Chemistry [English] Class 10 ICSE chapter 6 - Electrolysis
Shaalaa.com has the CISCE Mathematics Concise Chemistry [English] Class 10 ICSE CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Selina solutions for Mathematics Concise Chemistry [English] Class 10 ICSE CISCE 6 (Electrolysis) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Selina textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Concise Chemistry [English] Class 10 ICSE chapter 6 Electrolysis are Preferential Or Selective Discharge of Ions at Electrodes, Examples of Electrolysis, Electrolysis of Molten Lead Bromid, Electrolysis of Acidified Water Using Platinum Electrodes, Electrolysis of Copper Sulphate Solution Using Platinum Anode and Copper Or Platinum Cathode, Electrolysis of Aqueous Copper Sulphate - Using Copper Electrodes, Applications of Electrolysis, Electrolysis, Electrolytes, Nonelectrolyte, Electrochemical Cells, Electrodes, Oxidation, Reduction and Redox Reactions, Arrhenius Theory of Electrolytic Dissociation, Electrochemical Series, Preferential Or Selective Discharge of Ions at Electrodes, Examples of Electrolysis, Electrolysis of Molten Lead Bromid, Electrolysis of Acidified Water Using Platinum Electrodes, Electrolysis of Copper Sulphate Solution Using Platinum Anode and Copper Or Platinum Cathode, Electrolysis of Aqueous Copper Sulphate - Using Copper Electrodes, Applications of Electrolysis, Electrolysis, Electrolytes, Nonelectrolyte, Electrochemical Cells, Electrodes, Oxidation, Reduction and Redox Reactions, Arrhenius Theory of Electrolytic Dissociation, Electrochemical Series.
Using Selina Concise Chemistry [English] Class 10 ICSE solutions Electrolysis exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Selina Solutions are essential questions that can be asked in the final exam. Maximum CISCE Concise Chemistry [English] Class 10 ICSE students prefer Selina Textbook Solutions to score more in exams.
Get the free view of Chapter 6, Electrolysis Concise Chemistry [English] Class 10 ICSE additional questions for Mathematics Concise Chemistry [English] Class 10 ICSE CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.
