Advertisements
Advertisements
प्रश्न
Define the following term:
Weak electrolytes
उत्तर
Electrolytes which allow small amount of electricity to flow through them and are partially dissociated in fused or aqueous solution are called weak electrolytes.
APPEARS IN
संबंधित प्रश्न
Name a non-metallic element which is a conductor of electricity.
Explain, why during the electrolysis of copper sulphate using copper electrodes, the colour of solution does not fade?
What is meant by the term 'electrolyte'?
What should be the physical state of lead bromide if it is to conduct electricity?
What particles are present in pure lead bromide?
Write down the word or phrase from the given options that will correctly fill in the blanks in the following sentence:
Pure water consists entirely of ________
Three different electrolytic cells, A, B and C are connected in separate circuits. Electrolytic cell A contains a sodium chloride solution. When the circuit is completed, a bulb in the circuit glows brightly. Electrolytic cell B contains an acetic acid solution and in this case, the bulb in the circuit glows dimly. The electrolytic cell C contains a sugar solution and the bulb does not glow. Give a reason for each of these observations.
Fill in the blank from the choices given below :
A molecule of _____ contains a triple bond.
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
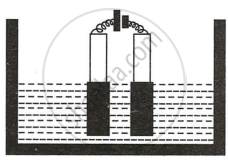
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Select the ion, that would get selectively discharge from the aqueous mixture of the ions listed below :
Pb2+, Ag+, Cu+
