Advertisements
Advertisements
प्रश्न
The electrolyte used for electroplating an article with silver is ______.
विकल्प
Silver nitrate solution
Silver cyanide solution
Sodium argentocyanide solution
Nickel sulphate solution
उत्तर
The electrolyte used for electroplating an article with silver is Sodium argentocyanide solution.
APPEARS IN
संबंधित प्रश्न
Give reasons why In the electroplating of an article with silver, the electrolyte sodium argentocyanide solution is preferred over silver nitrate solution.
How would you change Cu2+ ion to Cu?
An electrolytic cell is set up using two platinum electrodes and an aqueous solution of copper (II) sulphate,
(a)draw a labelled diagram of the electrolytic cell,
(b)Name the ions present in the cell,
(c)Name the ions migrating towards the anode,
(d)Name the ions migrating towards the cathode,
(e)Name the ions which will not be discharged at electrodes during electrolysis,
(f)Write the reaction at the cathode,
(g)Write the reaction at the anode,
(h)Name the spectator ion in the solution.
Explain the terms : Electro refining
List out the main applications of electrolysis.
Element X is a metal with valency 2. Element Y is a non-metal with valency 3.
(a) Write equations to show how X and Y form ions.
(b) If Y is diatomic gas, write the equation for the direct combination of X and Y to form a compound.
(c) Write two applications of electrolysis in which anode diminish in mass.
(d) If the compound formed between X and Y is melted and an electric current passed through the molten compound, the element X will be obtained at the _____ and the Y at the ________of the electrolytic cell. (Provide the missing words).
Copy and complete the following table :
| Anode | Electrolyte | |
| Purification of copper |
Describe what happens to the iron object and the copper rod.
Write two applications of electrolysis in which the anode diminishes in mass.
Electroplating steel objects with silver involves a three-step process.
Step 1: A coating of copper is applied to the object.
Step 2: A coating of nickel is applied to the object.
Step 3: The coating of silver is applied to the object.
-
- A diagram of the apparatus used for step 1 is shown
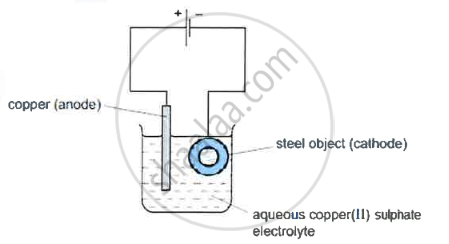
- The chemical process taking place on the surface of the object is \[\ce{Cu^2+(aq) + 2e- ->Cu(s)}\]
What is the observation seen on the surface of the object? - Explain why the concentration of copper ions in the electrolyte remains constant throughout step 1.
- The chemical process taking place on the surface of the object is \[\ce{Cu^2+(aq) + 2e- ->Cu(s)}\]
- A diagram of the apparatus used for step 1 is shown
- Give two changes which would be needed in order to coat nickel on to the object in step 2.
- Write down the reaction taking place at the positive electrode during step 3.
