Advertisements
Advertisements
प्रश्न
Electroplating steel objects with silver involves a three-step process.
Step 1: A coating of copper is applied to the object.
Step 2: A coating of nickel is applied to the object.
Step 3: The coating of silver is applied to the object.
-
- A diagram of the apparatus used for step 1 is shown
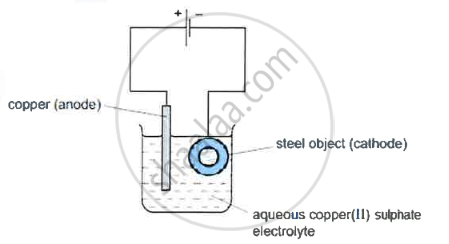
- The chemical process taking place on the surface of the object is \[\ce{Cu^2+(aq) + 2e- ->Cu(s)}\]
What is the observation seen on the surface of the object? - Explain why the concentration of copper ions in the electrolyte remains constant throughout step 1.
- The chemical process taking place on the surface of the object is \[\ce{Cu^2+(aq) + 2e- ->Cu(s)}\]
- A diagram of the apparatus used for step 1 is shown
- Give two changes which would be needed in order to coat nickel on to the object in step 2.
- Write down the reaction taking place at the positive electrode during step 3.
उत्तर
-
- reddish brown deposit/pink deposit/mass increases
- As anode released Copper ions the concentration of copper ions does not decrease
- The anode should be made up of Nickel and the electrolyte should be aq. Nickel sulphate or any salt solution of Nickel
- \[\ce{Ag -> Ag+ + e-}\]
APPEARS IN
संबंधित प्रश्न
State one relevant observation for the following:
At the anode when aqueous copper sulphate solution is electrolysed using copper electrodes.
Copy and complete the following table:
| Anode | Electrolyte | |
| Purification of copper |
Give one word or phrase for the following:
Electrolytic deposition of a superior metal on a baser metal.
State the electrode reaction at the anode during electrolysis of:
Aqueous copper sulphate solution.
Give reason for the following:
The blue colour of aqueous copper sulphate fades when it is electrolysed using platinum electrodes.
Explain the terms : Electro refining
The following question relate to the electroplating of an article with silver.
What ions must be present in the electrolyte?
Copy and complete the following table :
| Anode | Electrolyte | |
| Purification of copper |
Draw a labelled diagram to show how iron is electroplated with copper.
Describe what happens to the iron object and the copper rod.
