Advertisements
Advertisements
प्रश्न
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
उत्तर
When Benzyl amine is treated with nitrous acid, firstly BDC is formed which is unstable and decomposes to Benzyl alcohol.
\[\ce{\underset{Benzyl amine}{C6H5CH2NH2} + HONO ->[HCl] C6\underset{Benzyl alcohol}{H5CH2OH} + N2 + H2O}\]
APPEARS IN
संबंधित प्रश्न
How do you convert the following: Ethanenitrile to ethanamine
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
What is the best reagent to convert nitrile to primary amine?
Suggest a route by which the following conversion can be accomplished.
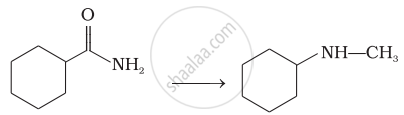
How will you carry out the following conversions?
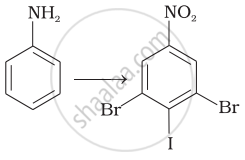
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
Write short note on the following.
Ammonolysis.
