Advertisements
Advertisements
प्रश्न
What is the shortest wavelength present in the Paschen series of spectral lines?
उत्तर
Rydberg’s formula is given as:
` "hc"/lambda = 21.76 xx 10^(-19) [1/"n"_1^2 - 1/"n"_2^2]`
Where,
h = Planck’s constant = 6.6 × 10−34 Js
c = Speed of light = 3 × 108 m/s
(n1 and n2 are integers)
The shortest wavelength present in the Paschen series of the spectral lines is given for values n1 = 3 and n2 = ∞.
`"hc"/lambda = 21.76 xx 10^(-19) [1/(3)^2 - 1/(∞)^2]`
`lambda = (6.6 xx 10^(-34) xx 3 xx 10^8 xx 9)/(21.76 xx 10^(-19)`
= 8.189 × 10−7 m
= 818.9 nm
APPEARS IN
संबंधित प्रश्न
An electron jumps from fourth to first orbit in an atom. How many maximum number of spectral lines can be emitted by the atom? To which series these lines correspond?
In both β− and β+ decay processes, the mass number of a nucleus remains the same, whereas the atomic number Z increases by one in β− decay and decreases by one in β+ decay. Explain giving reason.
Determine the series limit of Balmer, Paschen, and Pfund series, given the limit for Lyman series is 912 Å.
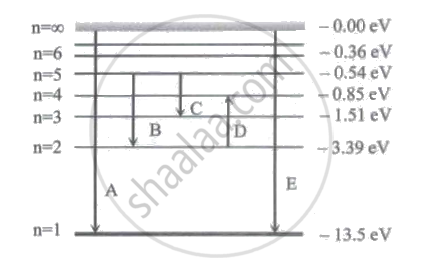
In the figure below, D and E respectively represent
Which of the following transition will have highest emission wavelength?
Let the series limit for Balmer series be 'λ1' and the longest wavelength for Brackett series be 'λ2'. Then λ1 and λ2 are related as ______.
Let v1 and v3 be the frequency for series limit of Balmer and Paschen series respectively. If the frequency of first line of Balmer series is v2 then, relation between v1 and v2 and v3 is ____________.
In hydrogen atom, the product of the angular momentum and the linear momentum of the electron is proportional to (n = principal quantum number) ____________.
If the mass of the electron is reduced to half, the Rydberg constant ______.
Each element is associated with a ______.
Show that the first few frequencies of light that is emitted when electrons fall to the nth level from levels higher than n, are approximate harmonics (i.e. in the ratio 1 : 2 : 3...) when n >> 1.
What is the minimum energy that must be given to a H atom in ground state so that it can emit an Hγ line in Balmer series. If the angular momentum of the system is conserved, what would be the angular momentum of such Hγ photon?
The first four spectral lines in the Lyman series of a H-atom are λ = 1218 Å, 1028Å, 974.3 Å and 951.4 Å. If instead of Hydrogen, we consider Deuterium, calculate the shift in the wavelength of these lines.
Determine the series limit of Balmer, Paschen and Brackett series, given the limit for Lyman series is 911.6 Å.
The frequencies for series limit of Balmer and Paschen series respectively are 'v1' and 'v3'. If frequency of first line of Balmer series is 'v2' then the relation between 'v1', 'v2' and 'v3' is ______.
In the hydrogen atoms, the transition from the state n = 6 to n = 1 results in ultraviolet radiation. Infrared radiation will be obtained in the transition.
