Advertisements
Advertisements
प्रश्न
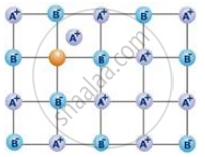
What type of defect is shown by NaCl in
stoichiometric defects, and
उत्तर

NaClshows vacancy or schottky defect in case of stoichiometric defect.
APPEARS IN
संबंधित प्रश्न
non-stoichiometric defects?
Answer the following question.
What type of stoichiometric defect is shown by ZnS and why?
Ionic solids containing large differences in sizes of ions show ____________.
Defect shown by non-ionic solids is ____________.
The following diagram shows:
Which is not correct about the Schottky defects?
Assertion: Due to Frenkel defect, there is no effect on the density of the crystalline solid.
Reason: In Frenkel defect, no cation or anion leaves the crystal.
Alkali halids do not show Frenkel defect because ____________.
Silver halides generally show:
Given below are two statements:
Statements I: Frenkel defects are vacancy as well as interstitial defects.
Statements II: Frenkel defect leads of colour in ionic solids due to the presence of F-centers.
Choose the most appropriate answer for the statements from the options given below:
