Advertisements
Advertisements
प्रश्न
What type of isomerism is exhibited by the complex [Co(en)3]3+?
(en = ethane-1,2-diamine)
उत्तर १
The complex [Co(en)3]3+ exhibits optical isomerism. Its optical isomers are shown below.
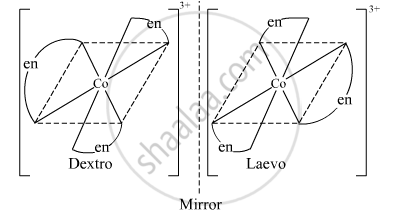
उत्तर २
Optical isomerism is exhibited by the complex [Co (en)3]3+.
APPEARS IN
संबंधित प्रश्न
Define the following term:
Anomers
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
[Pt(NH3)(H2O)Cl2]
How many geometrical isomers are possible in the following coordination entity?
[Cr(C2O4)3]3−
Draw the structure of optical isomers of [Cr(NH3)2Cl2(en)]+.
Draw all the isomers (geometrical and optical) of [Co(NH3)2Cl2(en)]+.
What type of structural isomers are [Co(NH3)5 Br] SO4 and [Co(NH3)5 SO4]Br? Give a chemical test to distinguish the isomers.
The IUPAC name for [CoCl(NO2)(en)2]Cl is ____________.
Which of the following molecules has a chiral centre correctly labelled with an asterisk (*)?
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers:
[Pt(NH3)(H2O)Cl2 ]
Assertion: Addition of bromine water to 1-butene gives two optical isomers.
Reason: The product formed contains two asymmetric carbon atoms.
