Advertisements
Advertisements
प्रश्न
When a mixture of gases X and Y is compressed to 300 atm pressure and then passed over a catalyst consisting of a mixture of zinc oxide and chromium oxide (heated to a temperature of 300°C), then an organic compounds Z having the molecular formula CH4O is formed. X is a highly poisonous gas which is formed in appreciable amounts when a fuel burns in a limited supply of air; Y is a gas which can be made by the action of a dilute acid on an active metal; and Z is a liquid organic compound which can react with sodium metal to produce hydrogen gas.
(a) What are X, Y and Z?
(b) Write a balanced chemical equation of the reaction which takes place when X and Y combine to form Z. Indicate the conditions under which the reaction occurs.
उत्तर
(a) X is carbon monoxide gas (CO); it is formed when a fuel burns in limited supply of air. Y is hydrogen gas (H2); it is formed by the action of dilute acid on active metal. Z is methanol (CH3OH); it is a liquid organic compound that can react with sodium metal to produce hydrogen gas.
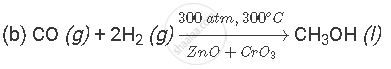
APPEARS IN
संबंधित प्रश्न
Fill in the following blank with suitable word:
A solution made in water is known as an ........... solution and indicated by the symbol ...........
Balance the following equation and add state symbols:
Zn + HCI → ZnCI2 + H2
Write the balanced chemical equation of the following reaction.
potassium dichromate + sulphuric acid → potassium sulphate + chromium sulphate + water + oxygen.
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of salt formed.
Give one example in the case where supplying energy [given below] is necessary for a chemical reaction.
Electrical energy
Name the following:
A catalyst which increases the rate of a chemical reaction.
\[\ce{2KClO3->[MnO2] 2KCl + 3O2[g] - is a balanced equation.}\]
Give a reason why the above equation is balanced.
Write a balanced equation for the following word equation:
Potassium bromide + Chlorine → Potassium chloride + Bromine
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Make a list of the elements in the chemical equation.
Complete the following blank in the equation as indicated.
\[\ce{CaH2_{(s)} + 2H2O_{( aq)}-> Ca(OH)2_{(s)} + 2H2_{(g)}}\]
Molecules: 6.02 × 1023 + ______ `→` ______ + ______
