Advertisements
Advertisements
प्रश्न
Which among the following is(are) double displacement reaction(s)?
- `"Pb" + "CuCl"_2 -> "PbCl"_2 + "Cu"`
- `"Na"_2"SO"_4 + "BaCl"_2 -> "BaSO"_4 + 2"NaCl"`
- `"C" + "O"_2 -> "CO"_2`
- `"CH"_4 + 2"O"_2 -> "CO"_2 + 2"H"_2"O"`
विकल्प
(i) and (iv)
(ii) only
(i) and (ii)
(iii) and (iv)
उत्तर
(ii) only
Explanation -
Only reaction (ii) is double displacement in nature.
APPEARS IN
संबंधित प्रश्न
On what basis is a chemical equation balanced?
Balance the given equation:
AI(OH)3  AI2O3 +H2O
AI2O3 +H2O
A metal X forms a salt XSO4. The salt XSO4 forms a clear solution in water which reacts with sodium hydroxide solution to form a blue precipitate Y. Metal X is used in making electric wire and alloys like brass.
(a) What do you think metal X could be?
(b) Write the name, formula and colour of salt XSO4.
(c) What is the blue precipitate Y?
(d) Write a chemical equation of the reaction which takes place when salt XSO4 reacts with sodium hydroxide solution. Give the state symbols of all the reactants and products which occur in the above equation.
Write word equation for the following skeletal equation:
\[\ce{Zn + HCl -> ZnCl2 + H2}\]
Balance the following chemical equation :
Na20 + H20 → NaOH
Balance the following equation:
NaHCO3 → Na2CO3 + H2O + CO2
Sodium chloride reacts with silver nitrate to produce silver chloride and sodium nitrate
Write the equation.
Check whether it is balanced, if not balance it.
Find the weights of reactants and products.
State the law which this equation satisfies.
Answer the following question:
How do acids and bases react with each other? What is the name of the process? What product is obtained from these reactions?
Write word equation for the following molecular equation:
\[\ce{NH3 [g] + HCl [g] ⇌ NH4Cl [s]}\]
Word equation:
State which characteristic is observed in the above reaction.
Give an-example of a similar above characteristic, seen between two other gaseous reactants.
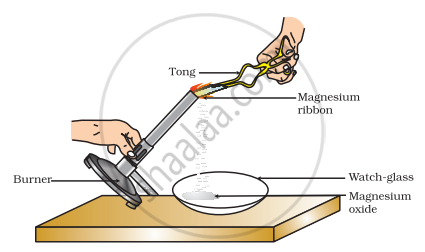
Which of the following is the correct observation of the reaction shown in the above set up?
