Advertisements
Advertisements
प्रश्न
Which electron displacement effect explains the following correct orders of acidity of the carboxylic acids?
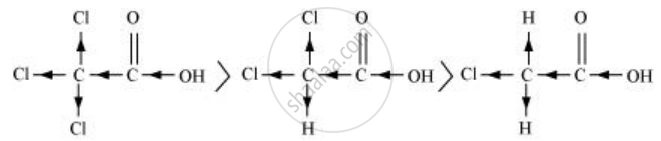
\[\ce{Cl3CCOOH > Cl2CHCOOH > ClCH2COOH}\]
टिप्पणी लिखिए
उत्तर
\[\ce{Cl3CCOOH > Cl2CHCOOH > ClCH2COOH}\]
The order of acidity can be explained on the basis of Inductive effect (–I effect). As the number of chlorine atoms increases, the –I effect increases. With the increase in –I effect, the acid strength also increases accordingly.
shaalaa.com
Fundamental Concepts in Organic Reaction Mechanism - Electron Displacement Effects in Covalent Bonds
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
अध्याय 12: Organic Chemistry - Some Basic Principles and Techniques - EXERCISES [पृष्ठ ३७१]
