Advertisements
Advertisements
प्रश्न
Which of the following diffuses fastest and which the slowest?
Solid, Liquid, Gas Give reasons for your answer.
उत्तर
Gases diffuse fastest and solids diffuse slowest when we compare solids, liquids and gases.
Explanation: Gases diffuse fast because the force of attraction between their particles is very low or negligible and there are vast spaces between the particles. The force of attraction between the particles of solids is very strong and there is no space between the particles due to their compact arrangement. As a result, rate of diffusion in solids is slowest.
APPEARS IN
संबंधित प्रश्न
Give a reason for the following observation.
Naphthalene balls disappear with time without leaving any solid.
State whether the following statement is true or false :
Red-brown bromine vapour diffuse into air in a gas jar but the colourless air molecules do not diffuse into bromine vapour.
Name two gases of air which dissolve in water by diffusion. What is the importance of this process in nature?
Why does a gas exert pressure?
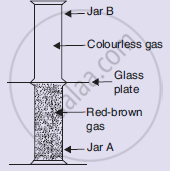
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
The space between ______ particles is the greatest one.
Comment upon the following:-
compressibility
Explain why?
- A gas fills a vessel completely?
- Camphor disappears without leaving any residue.
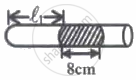 |
|
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)
A gaseous mixture of He and O2 is found to have a density of 0.518 gL-1 at 25° C and 720 torr. The mass percent of helium in this mixture is ______.