Advertisements
Advertisements
प्रश्न
Which of the following diffuses fastest and which the slowest?
Solid, Liquid, Gas Give reasons for your answer.
उत्तर
Gases diffuse fastest and solids diffuse slowest when we compare solids, liquids and gases.
Explanation: Gases diffuse fast because the force of attraction between their particles is very low or negligible and there are vast spaces between the particles. The force of attraction between the particles of solids is very strong and there is no space between the particles due to their compact arrangement. As a result, rate of diffusion in solids is slowest.
APPEARS IN
संबंधित प्रश्न
Tabulate the differences in the characteristics of states of matter.
Which of the two diffuses faster : a liquid or a gas?
Give two reasons to justify that Water is a liquid at room temperature.
What does the diffusion of gases tell us about their particles?
When a gas jar full of air is placed upside down on a gas jar full of bromine vapours, the red-brown vapours of bromine from the lower jar go upward into the jar containing air. In this experiment :
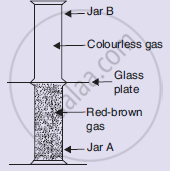
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
The space between ______ particles is the greatest one.
Comment upon the following:-
filling a gas container
Which of the following is TRUE about gaseous state?
2 litre He gas at 2 atm and 300 K is inserted into a 4 litre rigid container containing N2 at 600 K and 4 atm. Finally mixture is maintained at 600 K temperature. The final pressure of gaseous mixture would be ______ torr.
