Advertisements
Advertisements
प्रश्न
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
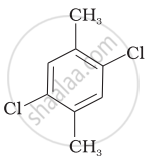 |
| (III) | 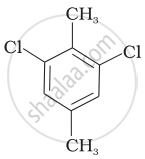 |
उत्तर

Compound (II)
Reasons: The molecular structure of compound (II) shows that both the substituents (i.e. \[\ce{CH3}\] and \[\ce{Cl}\] are at para position. The para-isomers have higher melting points as compared to their ortho and meta-isomers. It is due to symmetry of para-isomers that fits in crystal lattice better as compared to ortho-and meta-isomers.
APPEARS IN
संबंधित प्रश्न
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Define racemic mixture.
Which of the following possesses the highest melting point?
Mg reacts with RBr best in ____________.
Which is the correct increasing order of boiling points of the following compounds?
1-Iodobutane, 1-Bromobutane, 1-Chlorobutane, Butane
Assertion: The boiling points of alkyl halides decrease in the order:
\[\ce{RI > RBr > RCl > RF}\]
Reason: The boiling points of alkyl chlorides, bromides and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
Why is the boiling point of o-dichlorobenzene higher than p-dichlorobenzene, but the melting point of para-isomer is higher than ortho-isomer?
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3 C(C2H5)2CH2Br}\]
