Advertisements
Advertisements
Question
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
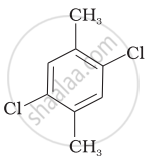 |
| (III) | 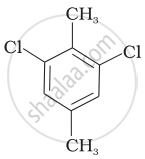 |
Solution

Compound (II)
Reasons: The molecular structure of compound (II) shows that both the substituents (i.e. \[\ce{CH3}\] and \[\ce{Cl}\] are at para position. The para-isomers have higher melting points as compared to their ortho and meta-isomers. It is due to symmetry of para-isomers that fits in crystal lattice better as compared to ortho-and meta-isomers.
APPEARS IN
RELATED QUESTIONS
Explain why alkyl halides, though polar, are immiscible with water?
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Which of the following compounds has the highest boiling point?
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
The decreasing order of boiling points of alkyl halides is:
Which of the following is liquid at room temperature (b.p. is shown against it)?
p-dichlorobenzene has a higher melting point than its o- and m-isomers because ____________.
Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
