Advertisements
Advertisements
प्रश्न
Which of the following is isoelectronic pair?
विकल्प
\[\ce{ICI2, ClO2}\]
\[\ce{BrO^{-}_{2}, BrF^{+}_{2}}\]
\[\ce{ClO2, BrF}\]
\[\ce{CN^{-}, O3}\]
उत्तर
\[\ce{BrO^{-}_{2}, BrF^{+}_{2}}\]
Explanation:
\[\ce{BrO^{-}_{2}}\] (35 + 2 × 8 + 1 = 52)
And
\[\ce{BrF^{+}_{2}}\] (35 + 2 × 9 – 1 = 52)
APPEARS IN
संबंधित प्रश्न
Arrange the following in the order of property indicated for each set:
HF, HCl, HBr, HI - increasing acid strength.
Complete the following equations:
KMnO4 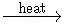
Arrange the following in the decreasing order of their reducing character :
HF, HCl, HBr, HI
Cl2 acts as a bleaching agent.
Tincture of iodine is ____________.
Fluorine differs from the rest of the halogens in some of its properties. This is due to ____________.
The set with the correct order of acidity is:
Which of the following statements are true?
(i) Only type of interactions between particles of noble gases are due to weak dispersion forces.
(ii) Ionisation enthalpy of molecular oxygen is very close to that of xenon.
(iii) Hydrolysis of XeF6 is a redox reaction.
(iv) Xenon fluorides are not reactive.
Solubility of iodine in water may be increase by adding
Write the name and formula of oxoacid of fluorine.
