Advertisements
Advertisements
प्रश्न
Which of the following statements are correct?
(i) Mixing two oppositely charged sols neutralises their charges and stabilises the colloid.
(ii) Presence of equal and similar charges on colloidal particles provides stability to the colloids.
(iii) Any amount of dispersed liquid can be added to emulsion without destabilising it.
(iv) Brownian movement stabilises sols.
उत्तर
(ii) Presence of equal and similar charges on colloidal particles provides stability to the colloids.
(iv) Brownian movement stabilises sols.
Explanation:
The presence of equal and similar charges on colloidal particles is largely responsible in providing stability to the colloidal solution, because the repulsive forces between charged particles having same charge prevent them from coalescing or aggregating when they come closer to one another. The Brownian movement has stirring effect which does not permit the particles to settle and thus, is responsible for the stability of sols.
APPEARS IN
संबंधित प्रश्न
Define the following terms: Associated colloids
Out of MgCl2 and AlCl3, which one is more effective in causing coagulation of negatively charged sol and why?
Define the following term : Brownian movement
Explain the terms Coagulation
What happens when a freshly prepared precipitate of Fe(OH)3 is shaken with a small amount of FeCl3 solution?
What happens when an emulsion is centrifuged?
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?
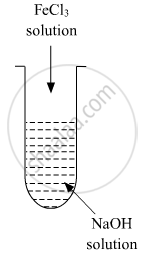
How does it become possible to cause artificial rain by spraying silver iodide on the clouds?
Define coagulation.
6.84 g Al2(SO4)3 is needed to coagulate 2.5 L of As2S3 sol completely in 2.0 hrs. The coagulation value of Al2 (SO4)3 is ______.
