Advertisements
Advertisements
प्रश्न
While deciding the unit for heat, which temperature interval is chosen? Why?
उत्तर
When deciding the unit for heat (which is a calorie), the temperature interval chosen is 14.5oC-15.5oC.
We know that the amount of heat released or absorbed by a body is given as `DeltaQ = msDeltaT`
Now, we also know that one calorie is defined as the amount of heat required to raise the temperature of 1 g of water through 1oC. Thus, for 1 calorie of heat energy, the specific heat capacity of water should be 1 cal g-1 oC-1. It is found experimentally that the specific heat capacity of water is 1 cal g-1 oC-1 when the temperature range is 14.5oC-15.5oC.
APPEARS IN
संबंधित प्रश्न
What is heat ? State its S.I. unit.
Name the mode of transfer of heat in the following :
(a) solid,
(b) liquid,
(c) gas
(d) vacuum
Draw a labelled diagram of a thermo flask. Explain how the transfer of heat by conduction, convection and radiation is reduced to a minimum in it.
Define the term heat.
What will absorb heat?
Glass
What will absorb heat?
Wooden spoon
What will absorb heat?
Plastic plate
What will absorb heat?
Soil
Complete the following sentence:
CGS units of heat is _________
The unit of heat and temperature are the same.
If you apply some heat energy to a substance, which of the following can take place in it?
A device in which the loss of heat due to conduction, convection and radiation is minimized is ______.
Ice is kept in a double-walled container. Why?
We feel the heat on our body when the ______ shines.
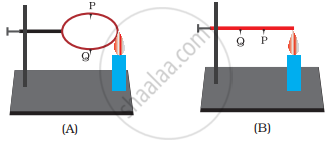
In arrangements, A and B shown in Figure 4.7, pins P and Q are fixed to a metal loop and an iron rod with the help of wax. In which case are both the pins likely to fall at different times? Explain.
The SI unit of heat energy is ______.
In summer, we prefer light – colored clothes and in winter we usually wear dark – colored clothes.
In which of the following, chemical energy is converted into heat energy?
Warm air is ______.
