Advertisements
Advertisements
Question
While deciding the unit for heat, which temperature interval is chosen? Why?
Solution
When deciding the unit for heat (which is a calorie), the temperature interval chosen is 14.5oC-15.5oC.
We know that the amount of heat released or absorbed by a body is given as `DeltaQ = msDeltaT`
Now, we also know that one calorie is defined as the amount of heat required to raise the temperature of 1 g of water through 1oC. Thus, for 1 calorie of heat energy, the specific heat capacity of water should be 1 cal g-1 oC-1. It is found experimentally that the specific heat capacity of water is 1 cal g-1 oC-1 when the temperature range is 14.5oC-15.5oC.
APPEARS IN
RELATED QUESTIONS
What is heat ? State its S.I. unit.
Draw a labelled diagram of a thermo flask. Explain how the transfer of heat by conduction, convection and radiation is reduced to a minimum in it.
Define the term calorie. How is it related to joule (the S.I. unit of heat)?
What will absorb heat?
Plastic plate
Give a scientific reason.
In winter, why does an iron pillar feel colder than a wooden pole?
Which temperature segment is chosen when determining the unit of heat? Why?
When an object is heated, the molecules that make up the object
______ energy can be generated by the burning of fuels like coal, wood, charcoal, gasoline, etc.
In the past, people used to rub two wooden pieces together to light fire.
A marble tile would feel cold as compared to a wooden tile on a winter morning because the marble tile
To keep her soup warm Paheli wrapped the container in which it was kept with a woollen cloth. Can she apply the same method to keep a glass of cold drink cool? Give a reason for your answer.
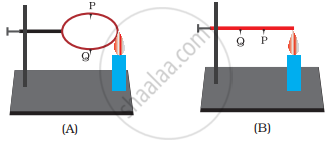
In arrangements, A and B shown in Figure 4.7, pins P and Q are fixed to a metal loop and an iron rod with the help of wax. In which case are both the pins likely to fall at different times? Explain.
The water in the black can becomes ______ than that in a white can after exposure to the sun.
Match the following
| 1. | Heat | a. | Good absorber |
| 2. | Temperature | b. | Form of energy |
| 3. | Black surface | c. | Insulators |
| 4. | Rubber, cork | d. | The measure of hotness or coldness |
Assertion: Small gaps left between railway lines.
Reason: It allows for the contraction of rails during summer.
Arrange the following in order of decreasing the expansion of heating: Steel, milk, air.
On a cold day, it is hard to open the lid of a tight container. But when you gently heat the neck you can easily open the lid. why?
