Advertisements
Advertisements
प्रश्न
While studying saponification reaction, a student measures the temperature of the reaction mixture and also finds its nature using blue/red litmus paper. On the basis of his observations the correct conclusion would be
(A) the reaction is exothermic and the reaction mixture is acidic.
(B) the reaction is endothermic and the reaction mixture is acidic.
(C) the reaction is endothermic and the reaction mixture is basic.
(D) the reaction is exothermic and the reaction mixture is basic.
उत्तर
Saponification reaction is a highly exothermic reaction. The reaction mixture of this reaction is basic in nature and hence, this will turn red litmus blue. Thus, it is basic in nature.
Hence, the correct option is D.
APPEARS IN
संबंधित प्रश्न
For preparing soap in the laboratory we require an oil and a base. Which of the following combinations of an oil and a base would be best suited for the preparation of soap?
(a) Castor oil and calcium hydroxide
(b) Turpentine oil and sodium hydroxide
(c) Castor oil and sodium hydroxide
(d) Mustard oil and calcium hydroxide
Complete the following chemical equations:CH3COOC2H5+NaOH→
You have four test tubes, A, B, C and D containing sodium carbonate, sodium chloride, lime water and blue litmus solutions respectively. Out of these the material of which test tube/ test tubes would be suitable for the correct test of acetic/ethanoic acid?
(a) only A
(b) A and B
(c) B and C
(d) A and D
Draw the structures for the following compounds of Propanoic acid.
Name the functional group present in the following compound:
HCOOH
What is vinegar and glacial acetic acid?
Identify the term or substance based on the descriptions given below:
Ice like crystals formed on cooling an organic acid sufficiently.
Which of the following represents saponification reaction?
In the presence of the acid catalyst, ethanoic acid reacts with ethanol and ______ ester is produced.
Observe the diagram given below and answer the questions:
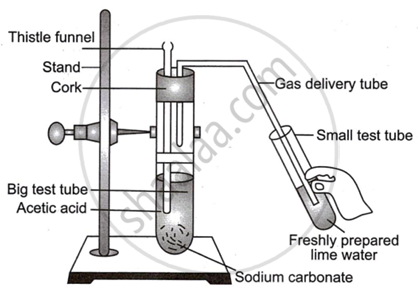
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
