Advertisements
Advertisements
प्रश्न
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
उत्तर
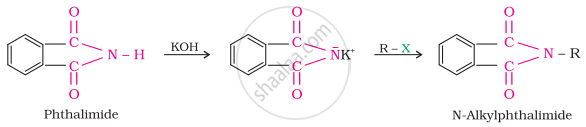
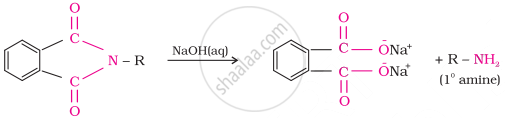
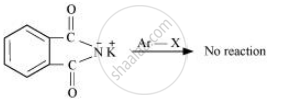
Gabriel synthesis is used for the preparation of primary amines. Phthalimide, on treatment with ethanolic potassium hydroxide, forms potassium salt of phthalimide, which on heating with alkyl halide followed by alkaline hydrolysis produces the corresponding primary amine. Aromatic primary amines cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide.
APPEARS IN
संबंधित प्रश्न
How do you convert the following: C6H5CONH2 to C6H5NH2
Write the reaction of aromatic primary amine with nitrous acid.
Give the structures of A, B and C in the following reactions :
Write structures of compounds A and B in each of the following reactions: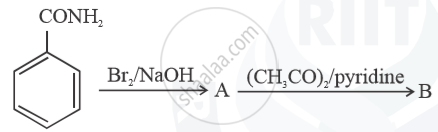
Answer the following
Explain the ammonolysis of alkyl halides.
Write reactions to prepare ethanamine from Acetonitrile.
Name the process of breaking C-X bond by ammonia in preparation of amines.
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Quaternary ammonium salt is formed:
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
Reduction of nitrobenzene by which of the following reagent gives aniline?
(i) \[\ce{Sn/HCl}\]
(ii) \[\ce{Fe/HCl}\]
(iii) \[\ce{H2 - Pd}\]
(iv) \[\ce{Sn/NH4OH}\]
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Which of the following compound is expected to be most basic?
What is the IUPAC name of \[\ce{(CH3)2 - N - CH3}\]?
Write short note on the following:
Ammonolysis
Write a short note on Ammonolysis.
Write a short note on the following:
Ammonolysis
