Advertisements
Advertisements
प्रश्न
Why is food containing iron salts should not be cooked in aluminium utensils?
उत्तर
Food containing iron salts should not be cooked in aluminium utensils as there is a danger of aluminium toxicity.
APPEARS IN
संबंधित प्रश्न
Aluminium is a more active metal than iron, but suffers less corrosion. Why?
Give reason for the following:
Nitric acid can be stored in aluminium containers.
How is ore purified (give equations also)
Write balanced equation for the following reaction:
Aluminium powder is warmed with hot and concentrated caustic soda solution.
The following question relate to the extraction of aluminium by electrolysis.
Give the equation for the reaction that takes place at the cathode
The following question relate to the extraction of aluminium by electrolysis.
Explain why is it necessary to renew the anode periodically.
Name the solution used to react with bauxite as a first step in obtaining pure aluminium oxide, in the Baeyer's process.
Write the balanced chemical equation to show the concentration of ore in Baeyer’s process.
Aluminium hydroxide to alumina
The following sketch illustrates the process of conversion of Alumina to Aluminium:
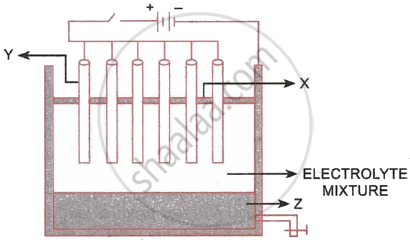
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
Given below in column A is a schematic diagram of the electrolytic reduction of alumina. Identify the parts labelled as A, B and C with the correct options from the Column B.
| column A | column B | |
 |
1. | Platinum |
| 2. | Anode | |
| 3. | Cathode | |
| 4. | Electrolyte mixture | |
| 5. | Bauxite |
