Advertisements
Advertisements
प्रश्न
With a labelled diagram describe an activity to show the formation of an ester.
उत्तर
1 mL of ethanol and 1 mL of glacial acetic acid are taken in a test tube. 3-4 drops of concentrated sulphuric acid are then added to a test tube. The resulting mixture in a water bath is then heated (as shown in the figure) for 5 minutes. After this, the contents are poured in a beaker containing 20 mL water. A sweet-smelling compound (ethyl ethanoalte) will be obtained as the product.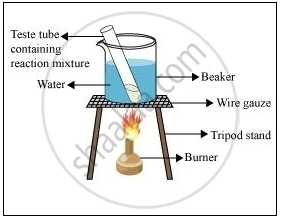
Formation of ester
When ethanoic acid reacts with ethanol in the presence of an acid, ethyl ethanoate is formed. Ethyl ethanoate is an ester and has a sweet smell.
APPEARS IN
संबंधित प्रश्न
Give a balanced chemical equation for Action of alcoholic KOH on bromethane
Fill in the following blank with suitable word:
The next higher homologue of ethane is ...............
Write the chemical equation of the reaction which takes place during the burning of ethanol in air.
Identify the functional group of the CH3OH ?
How does ethanol reacts with
(a) Metallic sodium
(b) Acetic acid
(c) Conc.H2S04
What are hydrocarbons? Give one example.
Generally _______ is called spirit.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
